Biologics Market Report Scope & Overview:
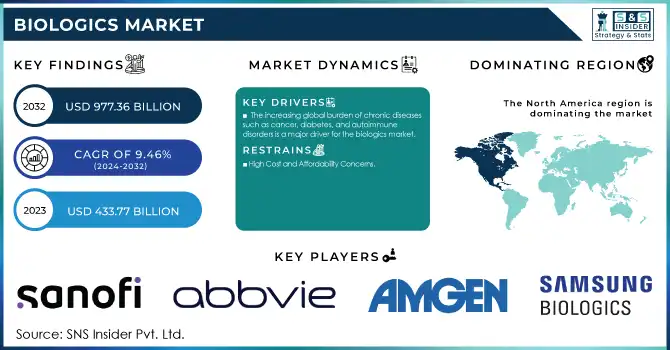
The Biologics Market size was valued at USD 519.72 Billion in 2025 and is projected to reach USD 1,283.29 Billion by 2035, growing at a CAGR of 9.46% during 2026–2035.
Biologics Market has gained prominence as an essential part of modern healthcare that provides innovative solutions to tackle health challenges. Various types of biologics such as monoclonal antibodies, vaccines, cell therapy/gene therapy, recombinant proteins, and RNA based therapies have been very effective in dealing with various chronic diseases like cancer, autoimmune diseases, and diabetes. Increasing occurrences of chronic illnesses, together with developments in biotechnological and genetic engineering technology, are driving the growth of this market during the forecasted years 2026 – 2035.
Keytruda (pembrolizumab), an immunotherapy for various cancers, has significantly improved survival rates in non-small cell lung cancer, while Humira continues to be a leading therapy for rheumatoid arthritis and Crohn's disease exemplifying the clinical impact and commercial scale that biologic medicines are delivering across oncology and autoimmune treatment globally.
To get more information on Biologics Market - Request Free Sample Report
Biologics Market Size and Forecast:
-
Market Size in 2025: USD 519.72 Billion
-
Market Size by 2035: USD 1,283.29 Billion
-
CAGR: 9.46% (from 2026 to 2035)
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
Biologics Market Highlights:
-
Rising prevalence of cancer, autoimmune disorders, and diabetes driving sustained demand for targeted biologic therapies globally
-
Monoclonal antibodies dominating the product landscape with approximately 45% market share, driven by oncology and immunology applications
-
Mammalian cell systems leading production with 65% share, enabling complex protein structures with human-like post-translational modifications
-
Antisense and RNAi therapeutics emerging as the fastest-growing product category driven by gene-level therapeutic targeting
-
Biosimilar expansion democratizing access to biologic therapies and driving market growth through improved affordability
-
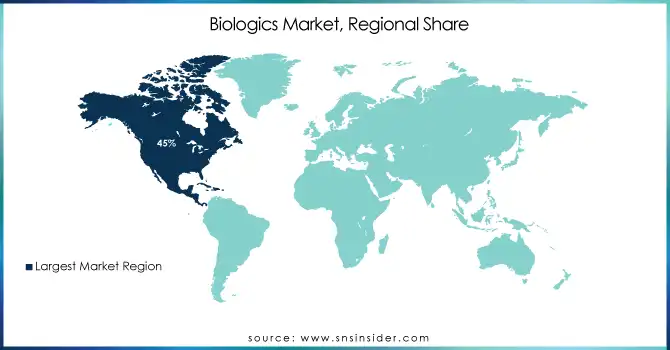
North America leads with 45% share in 2025, while Asia-Pacific is expected to register the fastest CAGR through 2026–2035
U.S. Biologics Market:
The U.S. Biologics Market was valued at USD 233.87 Billion in 2025 and is expected to reach USD 577.48 Billion by 2035, growing at a CAGR of 9.46% from 2026–2035.
Biologics research and development are witnessing an increase in the United States because of FDA approval, high demand for targeted drugs, adequate research and development funding, and constant advancement in biotech platforms. Major players like Amgen, Genentech, and Bristol-Myers Squibb have increased their drug pipelines and are making rapid strides in their clinical development.
Biologics Market Segment Highlights:
-
By Source: Mammalian Cells (Dominant – 65% share in 2025); Microbial Cells (Fastest Growing through 2026–2035)
-
By Product: Monoclonal Antibodies (Dominant – ~45% share in 2025); Antisense & RNAi Therapeutics (Fastest Growing through 2026–2035)
-
By Disease Category: Oncology (Dominant); Autoimmune Disorders (Fastest Growing through 2026–2035)
-
By Manufacturing: Contract Manufacturing (Dominant historically); In-House (Fastest Growing as firms seek greater control through 2026–2035)
Biologics Market Segment Analysis:
By Source, Mammalian Cells segment dominates the Biologics Market, Microbial Cells segment expected to grow fastest.
Mammalian cells dominated Biologics Market, supported by their capability to create complex, human-type proteins having accurate post-translational modifications, which are required to make the drug effective and less immunogenic. Popular mammalian cells like CHO cells can be used to create monoclonal antibodies, recombinant proteins, and fusion proteins on an industrial scale. This, along with their reliability and ease of integration in biologics manufacturing processes, helps to maintain their dominance in the industry.
Microbial cells are emerging as the fastest-growing segment through 2026–2035, driven by their cost-effectiveness and fast reproduction rates. This is especially efficient when manufacturing less complex biological compounds like insulin, vaccines, and growth hormones. The need for low-cost biological substances and developments in genetic engineering are further fueling the trend towards using microbes as production platforms.
By Product, Monoclonal Antibodies segment dominates the Biologics Market, Antisense & RNAi Therapeutics segment expected to grow fastest.
Monoclonal antibodies dominated the biologics market, driven by their high specificity, high efficacy, and wide-ranging applicability in the fields of oncology and immunology. The specific targeting ability of monoclonal antibodies has played a major role in ensuring their effectiveness in treating diseases. Furthermore, the continuous research and development of antibodies such as bispecific antibodies and antibody-drug conjugates will play an important role in sustaining their dominant market position.
Antisense and RNAi drugs will experience the highest CAGR over 2026–2035, thanks to the fast development of gene therapies based on targeting RNA molecules. This approach allows modulating gene expression and creating innovative treatment solutions for genetic diseases that cannot be treated by conventional means. The rapid progress in developing antisense drugs and growing use of RNA-based therapies across a number of therapeutic areas fuel market growth.
By Disease Category, Oncology segment dominates the Biologics Market, Autoimmune Disorders segment expected to grow fastest.
Oncology held a leading share of the market owing to the large number of patients suffering from various types of cancers across the globe and the ever-increasing development of targeted drugs, biologicals, and personalized medicine. Increased investments in research and clinical trials along with advanced modes of therapy are contributing to increased production and marketing of products in this field.
Autoimmune diseases are expected to register the highest CAGR through 2026-2035, owing to the growing incidence of chronic auto-immune diseases and increasing knowledge about the importance of early diagnosis and treatment. Biologic, biosimilar, and targeted immunotherapy treatments are proving more efficacious and providing greater access to patients.
By Manufacturing, Contract Manufacturing segment dominates the Biologics Market, In-House segment expected to grow fastest.
Contract manufacturing dominated the market, due to outsourcing of production activities by pharmaceutical and biotech firms to contract manufacturers in order to save on costs and ensure faster delivery of products. These manufacturers have high levels of expertise in terms of capabilities and flexibility of production, which makes them more appealing.
In-house manufacturing is anticipated to register the highest growth rate until 2026–2035, driven by increasing concerns about the quality of production, proprietary information protection, and supply chain security. Companies have been making significant investments in advanced manufacturing technologies and systems, thereby allowing for process internalization. This trend is especially noticeable in high-end products such as biologics and personalized treatments.
Biologics Market Regional Analysis:
|
Region |
Major Country |
Share (%) |
|---|---|---|
|
North America |
United States |
75.0% |
|
Europe |
Germany |
23.0% |
|
Asia Pacific |
China |
20.0% |
|
Middle East & Africa |
Saudi Arabia |
6.0% |
|
Latin America |
Brazil |
6.0% |
North America Biologics Market Insights:
The Biologics Market was led by North America, which accounted for around 45% revenue share in 2025 owing to its advanced biopharmaceutical industry, well-developed healthcare facilities, high spending on research & development, conducive regulatory landscape, and availability of major biopharmaceutical players globally. The United States is at the forefront of biologic drugs development, with key players such as Amgen Inc., Genentech Inc., and Bristol Myers Squibb Co. playing an instrumental role in the advancement of antibody technology, gene-based therapies, and innovative bioprocessing techniques.
In January 2025, the U.S. FDA continued to accelerate its biosimilar approval pathway, with over 40 biosimilar products approved in the U.S. by early 2025—expanding patient access, driving competition, and creating significant market opportunities for both innovator biologics and biosimilar manufacturers across North America.
Asia-Pacific Biologics Market Insight:
The Asia-Pacific region is expected to be the fastest growing biologics market during the period of 2026-2035 owing to various reasons including healthcare infrastructure development, increased disease burden, enhanced capabilities in biopharmaceutical manufacturing and supportive government policies for indigenous biologics manufacture. Emerging markets of China and India are expected to play an increasingly important role in terms of being the producers and consumers of biologics owing to their substantial investment in the biotechnology industry as well as enhanced clinical research efforts.
China's National Medical Products Administration has streamlined biologics approval pathways and established favorable policies for domestic biopharmaceutical development, encouraging substantial private sector investment in biologic drug pipelines targeting oncology, metabolic diseases, and rare genetic disorders positioning China as a rapidly emerging force in global biologics innovation and production.
Europe Biologics Market Insights:
Europe plays an important role in the Biologics Market due to its highly developed biopharmaceutical industry coupled with the presence of effective regulatory authorities in the form of the European Medicines Agency, along with extensive use of biologics within its specialty areas such as oncology, rheumatology, and orphan drugs. Europe has one of the best biosimilars markets worldwide, owing to the existence of a reliable biosimilar drug development process approved by the EMA which allows for competitive pricing. Contributing countries in the region include Germany, Switzerland, UK, and Denmark.
The European Medicines Agency has approved over 90 biosimilar medicines as of 2024 more than any other regulatory body globally—demonstrating Europe's leadership in biosimilar policy and its role as a global model for expanding patient access to biologic therapies through regulatory-supported market competition.
Middle East & Africa and Latin America Biologics Market Insights:
The Middle East and Africa and Latin America demonstrated growing biologics market participation in 2025, due to higher chronic disease prevalence, better healthcare facilities, and increased government spending to widen the reach of new biologic drugs. Brazil, Saudi Arabia, and the United Arab Emirates have been the key market players responsible for market growth in both geographies. Increased usage of biosimilars has helped make biologics more widely available. Governments have made efforts to encourage local drug manufacturing and cancer drug therapy availability.
Biologics Market Drivers:
-
Rising prevalence of chronic diseases drives sustained demand for targeted and personalized biologic therapies globally
The increasing burden worldwide due to various chronic ailments such as cancer, autoimmune diseases, diabetes, and various genetic conditions is the major structural force behind the Biologics Market. Often, such diseases necessitate the need for highly advanced treatments that small molecule drugs do not offer, thus presenting a strong case for the use of biological drugs, which can specifically target various biological and genetic pathways. The World Health Organization estimates that 74% of deaths globally result from chronic conditions, along with an increase in patients in developed and developing countries, thereby driving demand in all product categories of biological drugs until 2035.
FDA granted approval to 55 new treatments in 2023, including 17 biologics with 12 being monoclonal antibodies—demonstrating the continued innovation pipeline and regulatory momentum that is sustaining strong commercial launches of new biologic therapies across oncology, immunology, and rare disease treatment categories globally.
-
Biosimilar expansion and personalized medicine advancement drive market growth and broad patient access to biologics
The fast growth of the Biologics market is ensuring increased accessibility to biologics treatments by reducing costs and introducing competition in areas that were protected under patents in the past. Regulatory agencies like FDA and EMA are assisting in facilitating rapid approval procedures for biosimilars, hence encouraging competition without compromising the efficacy and safety of such treatments. At the same time, the rising interest in precision medicine and companion diagnostics is ensuring that there is increased targeting of patients, thereby improving effectiveness and allowing greater viability of biologics in difficult-to-treat conditions.
Biologics Market Restraints:
-
High development costs, complex manufacturing requirements, and lengthy regulatory processes limit market entry and increase therapeutic costs
The development of biological drugs involves a huge expenditure on bioprocessing equipment, cell lines, clinical testing, and regulatory affairs, all of which far exceed the expenditure incurred in the development of small molecule drugs. The high costs associated with the process of bioprocessing, cold storage of products, and batch-to-batch variations in products produced using living cells are some of the reasons for higher costs per unit that result in premium prices. Such considerations, coupled with the lengthy period of regulatory approval, which may take up to ten years from the time of discovery to commercialization, pose a big barrier to market entry. This advantage is usually enjoyed by well-established large pharma firms but not by smaller biotech firms.
Biologics Market Opportunities:
-
Cell & Gene Therapy Breakthrough Applications and Emerging Market Biosimilar Expansion Create Transformative Growth Opportunities
The cell and gene therapy arena constitutes one of the most promising commercially-oriented areas of growth within the Biologics Market, as revolutionary treatments with potential to cure otherwise unapproachable genetic and oncological diseases generate unprecedented interest among investors and patients. The approval process of CAR-T cell treatments and gene substitution therapy is paving the way to demonstrating the profitability of such advanced biological approaches. Moreover, the immense demand for inexpensive biologics in Asia-Pacific, Latin American, and Middle Eastern regions provides substantial business opportunities for companies developing biosimilars and undertaking technology transfer programs.
Get Customized Report as per Your Business Requirement - Enquiry Now
Recent Developments:
-
In January 2025, the FDA approved a new biosimilar to Humira (adalimumab), expanding the competitive biosimilar landscape for one of the world's best-selling biologic drugs and increasing patient access to affordable treatment options for rheumatoid arthritis and Crohn's disease.
-
In 2024, Amgen launched Blincyto (blinatumomab) as a subcutaneous formulation, improving patient convenience for this bispecific T-cell engager used in acute lymphoblastic leukemia treatment and demonstrating continued innovation in biologic drug delivery.
-
In 2023, the FDA approved Leqembi (lecanemab), a monoclonal antibody developed by Eisai and Biogen for early Alzheimer's disease treatment, marking a significant milestone in the development of biologic therapies for neurological conditions.
-
In 2022, BioNTech announced expansion of its mRNA biologic pipeline beyond COVID-19 vaccines to include oncology applications, signaling a transformative broadening of RNA-based biologic technology into new high-value therapeutic categories.
Biologics Market Key Players:
-
Samsung Biologics
-
Amgen Inc.
-
AbbVie Inc.
-
Novo Nordisk A/S
-
F. Hoffmann-La Roche Ltd.
-
Pfizer Inc.
-
Sanofi S.A.
-
Johnson & Johnson Services, Inc.
-
Celltrion Healthcare Co., Ltd.
-
Bristol-Myers Squibb Company
-
Eli Lilly and Company
-
Merck & Co., Inc.
-
AstraZeneca plc
-
Gilead Sciences, Inc.
-
Novartis AG
-
Regeneron Pharmaceuticals, Inc.
-
Takeda Pharmaceutical Company
-
Biogen Inc.
-
Genentech (Roche)
-
CSL Behring
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 519.72 Billion |
| Market Size by 2035 | USD 1283.29 Billion |
| CAGR | CAGR of 9.46% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Source [Microbial, Mammalian, Others] • By Product [Monoclonal Antibodies {MABs by Application (Diagnostic {Biochemical Analysis, Diagnostic Imaging}, (Therapeutic {Direct MAB Agents, Targeting MAB Agents (Protein Purification, Others)}, {MABs by Type (Murine, Chimeric, Humanized, Human, Others)}, Vaccines, Recombinant Proteins, Antisense & RNAi Therapeutics, Others] • By Disease Category [Oncology, Infectious Diseases, Immunological Disorders, Cardiovascular Disorders, Hematological Disorders, Others] • By Manufacturing [Outsourced, In-house] |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
| Company Profiles |
Samsung Biologics, Amgen Inc., AbbVie Inc., Novo Nordisk A/S, F. Hoffmann-La Roche Ltd., Pfizer Inc., Sanofi S.A., Johnson & Johnson Services, Inc., Celltrion Healthcare Co., Ltd., Bristol-Myers Squibb Company, Eli Lilly and Company, Merck & Co., Inc., AstraZeneca plc, Gilead Sciences, Inc., Novartis AG, Regeneron Pharmaceuticals, Inc., Takeda Pharmaceutical Company, Biogen Inc., Genentech (Roche), CSL Behring |

