Medical Nonwoven Disposable Market Report Scope & Overview:
Get more information on Medical Nonwoven Disposable Market - Request Sample Report
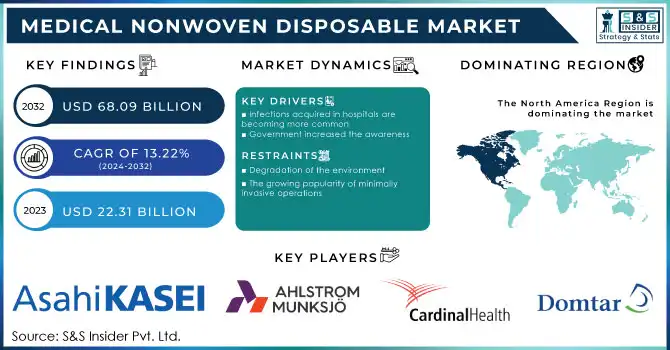
The Medical Nonwoven Disposable Market was valued at USD 27.76 billion in 2023 and is expected to reach USD 75.29 billion by 2032, growing at a CAGR of 11.77% from 2024-2032.
The medical nonwoven disposable market report provides unique insights into leading trends and drivers influencing the industry. It delivers a thorough analysis of market demand by product type, including the specific demand for surgical gowns, wound care products, face masks, and others. The report further examines regional patterns of adoption of nonwoven disposable products, offering geographic differences and drivers of demand by region. Sustainability trends are also examined, highlighting the growing importance of eco-friendly, biodegradable nonwoven solutions to lower the environmental impact of disposable medical products. Furthermore, the report identifies the rise of home healthcare, describing how the expansion of home care and the development of nonwoven technology are driving market growth between 2023 and 2032.
Market dynamics
Drivers
-
The rising emphasis on infection prevention and control in hospitals and clinics is driving the adoption of medical nonwoven disposables such as surgical gowns, drapes, and masks.
The growing focus on infection prevention and control in hospitals and clinics is the driving factor behind increasing demand for medical nonwoven disposables such as surgical gowns, drapes, and masks. Rising awareness about HAIs and increasing infection control measures have accelerated the adoption of disposable products for ensuring hygiene and patient safety. For example, the CDC estimates that around 1 in 31 patients who come to the hospital carry at least one healthcare-associated infection, which has further emphasized the need for disposable products. The recent developments are about nonwoven materials that can be used as antimicrobial and fluid-repellent, ensuring increased protection for health care professionals and patients. Disposable products especially for surgical and ICU environments have been introduced by manufacturers such as 3M and Kimberly-Clark; this has increased market demand.
-
The growing aging population and a shift toward home healthcare have significantly boosted the demand for medical nonwoven disposables, including adult diapers, incontinence products, and wound care materials.
A growing aging population and a shift toward home healthcare have greatly increased the demand for medical nonwoven disposable products, such as adult diapers, incontinence products, and wound care materials. The WHO expects that, by 2050, the world population aged 60 and above will double, creating a massive need for these products. This has been further increased with the upsurge of chronic diseases like diabetes and incontinence with continuous requirements of nonwoven disposables at home care facilities. The product line across Domtar Corporation and Essity with biodegradable properties is also enhancing the product lines under the requirement of the patient and the environmental concerns.
Restraint
-
Environmental Concerns and Waste Management Challenges Restraining the Medical Nonwoven Disposable Market
The increasing use of disposable medical nonwoven products is posing significant environmental challenges due to the accumulation of medical waste. Nonwoven disposables, primarily made from synthetic materials like polypropylene, contribute to non-biodegradable waste, raising concerns about their long-term environmental impact. According to the WHO, approximately 85% of the total medical waste generated globally is non-hazardous but largely disposable. This has led to growing scrutiny from environmental agencies and a push for sustainable alternatives. Though efforts are being made to develop biodegradable nonwoven materials, higher production costs and limited availability of eco-friendly options are hindering their widespread adoption, thus restraining market growth.
Opportunities
-
Rising Demand for Biodegradable and Sustainable Nonwoven Products benefiting Medical Nonwoven Disposable Market
The increasing global focus on environmental sustainability is a significant opportunity for the medical nonwoven disposable market. As plastic usage is increasingly regulated and consumer awareness increases, healthcare providers are looking for eco-friendly alternatives to traditional nonwoven products. Biodegradable and compostable nonwovens, such as those made from natural fibers like bamboo and cotton, offer a promising solution. Examples include, but are not limited to plant-based surgical gowns, or biodegradable wound dressings. All the companies can attract this potential green demand in line with improving nonwoven-related market positions for them.
Challenges
-
One of the significant challenges in the medical nonwoven disposable market is the high cost associated with advanced nonwoven products.
One of the major challenges facing the medical nonwoven disposable market is that advanced nonwoven products are very expensive. Although these materials have superior performance, such as enhanced absorbency, antimicrobial properties, and biodegradability, they are usually much more expensive than traditional products. This cost factor limits their adoption, especially in price-sensitive markets and smaller healthcare facilities. Other factors, that contribute to high costs, are specialized manufacturing technologies and raw materials for these products. Overcoming this challenge calls for innovative production processes and economies of scale in the production of sophisticated nonwoven products to make them more affordable and widely available.
Segmentation Analysis
By Product
The medical supplies segment dominated the market in 2023 due to the critical role of nonwoven fabrics in the wide range of essential medical applications. Nonwoven materials are ideal for the manufacture of such products as surgical drapes, gowns, wound care products, face masks, and other disposable supplies for hygiene and infection control in health care. They are lightweight, inexpensive, and flexible, which makes them suitable for hospitals, clinics, and surgeries.
Moreover, an increased focus on the prevention of infection and hygiene in medical settings was another factor that raised the market for medical supplies. Improved fluid barrier properties and better breathability of nonwoven technology further enhanced the functionality and reliability of these products. This resulted in the largest market share of the medical supplies segment in 2023 since healthcare facilities continued to put high investments into disposable nonwoven products for safety, regulatory, and patient care requirements.
Regional Analysis
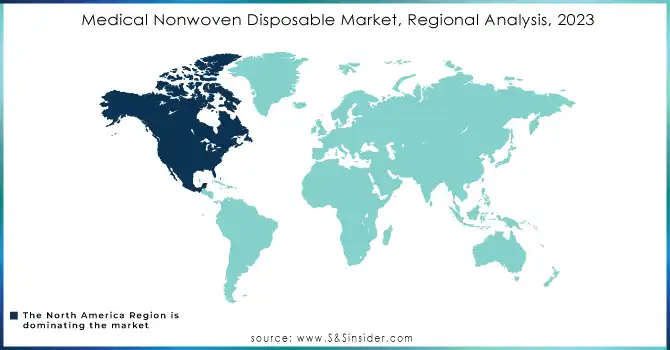
North America dominated the market with a 38.46% market share in 2023, because of its well-developed healthcare infrastructure, high spending on healthcare, and strict regulatory standards that encourage the use of advanced medical products. The growing geriatric population and the increasing incidence of chronic diseases, such as diabetes and cardiovascular disorders, have fueled the demand for disposable medical products, including wound care supplies, surgical gowns, and face masks. In addition, North America features a strong manufacturing network. Several key players are investing in innovative solutions and expanding their portfolios. Thus, the region maintains its predominant position. Healthcare providers in the United States and Canada demand excellent, high-reliability nonwovens for providing absolute patient safety with the most effective medical outcome.
Asia Pacific region experiencing the fastest growth with 14.79% CAGR throughout the forecast period, mainly driven by rapid economic growth, increasing healthcare investment, and a rising population with evolving healthcare needs. Countries like China, India, and Japan are witnessing a surge in demand for nonwoven products due to expanding healthcare infrastructure, awareness of hygiene, and growing infections and chronic conditions. Technological advancements and health insurance coverage expansion in the region's healthcare sector are helping improve its overall condition, making medical nonwoven disposables more accessible. With the increasing urbanization rate and fast growth of the healthcare modernization process, an increased demand for disposable medical products is likely to be witnessed, propelling the Asia Pacific market rapidly.
Need any customization research on Medical Nonwoven Disposable Market - Enquiry Now
Some of the major key players in the Medical Nonwoven Disposable Market
-
3M Company (N95 Respirators, Surgical Drapes)
-
Kimberly-Clark Corporation (Disposable Surgical Gowns, Incontinence Products)
-
Mölnlycke Health Care (Procedure-Specific Surgical Packs, Wound Care Dressings)
-
Berry Global Group, Inc. (Disposable Face Masks, Medical Wipes)
-
Cardinal Health, Inc. (Surgical Drapes and Gowns, Disposable Patient Apparel)
-
Medline Industries, Inc. (Disposable Bed Pads, Isolation Gowns)
-
DuPont de Nemours, Inc. (Tyvek Protective Apparel, Sterile Disposable Gowns)
-
Ahlstrom-Munksjö (Medical Sterilization Wraps, Face Mask Materials)
-
Asahi Kasei Corporation (Surgical Face Masks, Disposable Aprons)
-
Freudenberg Performance Materials (Biodegradable Nonwoven Wound Dressings, Single-Use Medical Wipes)
-
Halyard Health (Sterile Surgical Gowns, Procedural Drapes)
-
Domtar Corporation (Adult Incontinence Products, Disposable Underpads)
-
TWE Group GmbH (Nonwoven Components for Wound Care, Disposable Surgical Materials)
-
Paul Hartmann AG (Absorbent Pads for Surgery, Disposable Face Masks)
-
Essity AB (Incontinence Pads, Disposable Sanitary Napkins)
-
KCWW (Kimberly-Clark Professional) (Disposable Gloves, Medical Gowns)
-
Hogy Medical Co., Ltd. (Nonwoven Surgical Drapes, Disposable Masks)
-
Advanced Fabrics (SAAF) (Single-Use Medical Wraps, Anti-Microbial Face Masks)
-
Fitesa (Nonwoven Surgical Gowns, Disposable Caps)
-
Ansell Limited (Sterile Disposable Gloves, Single-Use Medical Aprons)
Suppliers (These suppliers play a critical role in providing raw materials and fabrics that are essential for manufacturing medical nonwoven disposable products.)
-
Berry Global Group, Inc.
-
Freudenberg Performance Materials
-
Ahlstrom-Munksjö
-
Fitesa
-
Pegas Nonwovens (PFNonwovens)
-
Toray Industries, Inc.
-
Johns Manville
-
Kimberly-Clark Professional
-
DuPont de Nemours, Inc.
-
Don & Low Ltd.
Recent development
-
November 2024, Berry Global Group, Inc. announced that its Health, Hygiene, and Specialties Global Nonwovens and Films business (HHNF Business) has completed its merger with Glatfelter Corporation. The result is Magnera Corporation, MAGN's largest nonwoven company in the world-offering an end-to-end solution to the specialty materials industry.
-
July 2023, Berry Global, in partnership with Deaconess Midtown Hospital, Nexus Circular, and Evansville Packaging Supply, has started a program to recycle sterile healthcare plastics, thereby reducing environmental waste. This collaboration aims to transform non-hazardous, sterile plastic packaging and nonwoven fabrics from surgical suites, pharmaceutical operations, laboratories, and warehouses.
| Report Attributes | Details |
|---|---|
| Market Size in 2023 | US$ 27.26 Billion |
| Market Size by 2032 | US$ 75.29 Billion |
| CAGR | CAGR of 11.77% From 2024 to 2032 |
| Base Year | 2023 |
| Forecast Period | 2024-2032 |
| Historical Data | 2020-2022 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | •By Product (Medical Supplies, Hygine Products) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | 3M Company, Kimberly-Clark Corporation, Mölnlycke Health Care, Berry Global Group, Inc., Cardinal Health, Inc., Medline Industries, Inc., DuPont de Nemours, Inc., Ahlstrom-Munksjö, Asahi Kasei Corporation, Freudenberg Performance Materials, Halyard Health, Domtar Corporation, TWE Group GmbH, Paul Hartmann AG, Essity AB, KCWW (Kimberly-Clark Professional), Hogy Medical Co., Ltd., Advanced Fabrics (SAAF), Fitesa, Ansell Limited, and other players. |

