Asthma Therapeutics Market Report Scope & Overview:
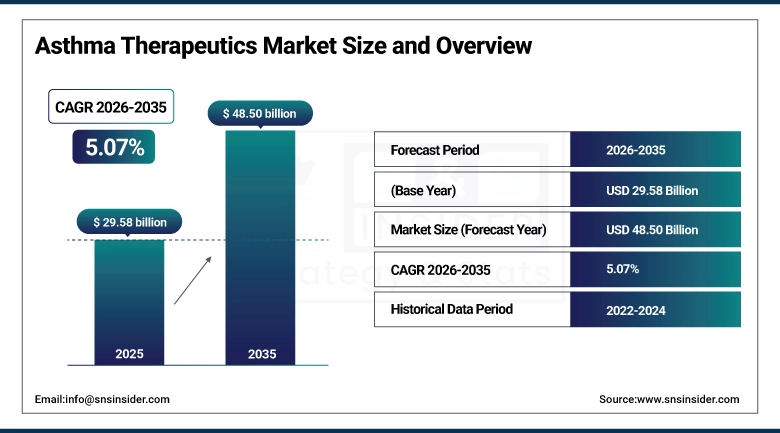
The Asthma Therapeutics Market size was valued at USD 29.58 billion in 2025 and is expected to reach USD 48.50 billion by 2035, growing at a CAGR of 5.07% over the forecast period of 2026–2035.
Key factors that are expected to drive the global market for asthma therapeutics include the rising prevalence of respiratory diseases, the technological advancements in inhalation devices, and the increasing number of biologics in the pipeline aimed at targets such as IgE, IL-4, IL-5, and IL-13. The World Health Organization (WHO) estimates that in 2019 about 262 million people across the globe were affected by the disease. To top it all off, it is responsible for more than 455,000 deaths across the globe. So clearly the condition is wonderfully managed, but there is growing need for solutions. The growing penetration of controller medications, such as inhaled corticosteroids and long-acting beta-agonists—coupled with an increasing recognition of the role that personalized medicine can play in enhancing the value of treatment—are redefining the market. Moreover, strong reimbursement environment in developed nations and increasing pipeline for biosimilars is propelling market potential in developed and low income nations.
For instance, in February 2024, the Global Initiative for Asthma (GINA) reported that less than 10% of asthma patients worldwide achieve optimal disease control, representing a critical unmet clinical need and a substantial commercial opportunity for pharmaceutical innovators and therapeutic device manufacturers.
Asthma Therapeutics Market Size and Forecast:
-
Market Size in 2025: USD 29.58 billion
-
Market Size by 2035: USD 48.50 billion
-
CAGR: 5.07% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get more information on Asthma Therapeutics Market - Request Free Sample Report
Asthma Therapeutics Market Trends:
-
Rapid adoption of biologic monoclonal antibody therapies, including anti-IL-5, anti-IgE, and anti-IL-4/IL-13 agents, is redefining treatment protocols for severe, refractory asthma unresponsive to conventional inhaled regimens.
-
Smart inhaler devices integrated with Bluetooth connectivity, dose counters, and real-time adherence tracking are gaining traction as healthcare systems prioritize measurable patient compliance outcomes.
-
Expansion of fixed-dose combination (FDC) inhaler formulations delivering ICS/LABA and ICS/LABA/LAMA triple therapy is reducing pill burden while significantly improving lung function and exacerbation rates.
-
Growing utilization of fractional exhaled nitric oxide (FeNO) testing and blood eosinophil counts as biomarker-driven patient stratification tools is driving precision prescribing across specialist and primary care settings.
-
Rising environmental pollution levels, urban heat exposure, and climate-driven allergen intensification are structurally elevating the global asthma disease burden, sustaining long-term demand for maintenance and rescue therapies.
-
Pediatric asthma therapeutics represent a distinct and expanding segment, with pharmaceutical companies investing in age-appropriate nebulizer solutions, low-dose ICS formulations, and child-friendly dry powder inhalers.
-
Increased regulatory focus on hydrofluorocarbon (HFC)-free and low-carbon propellant inhalers, particularly in Europe, is accelerating the transition toward next-generation environmentally sustainable inhaler platforms.
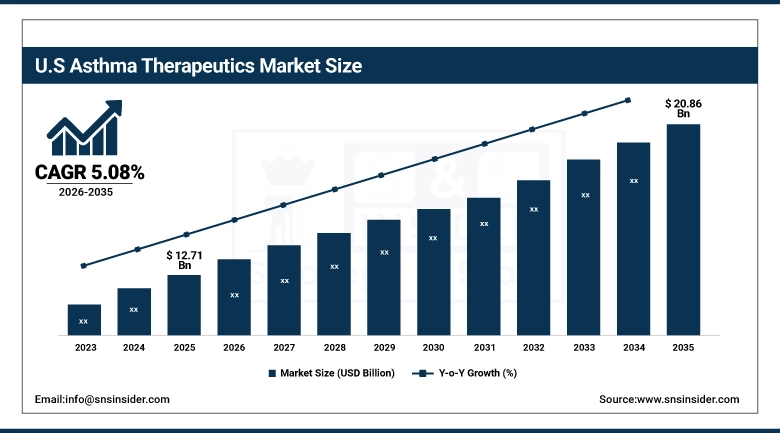
The U.S. Asthma Therapeutics Market was valued at approximately USD 12.71 billion in 2025 and is projected to reach USD 20.86 billion by 2035, growing at a CAGR of 5.08% from 2026 to 2035. The United States leads in terms of national market share in the global asthma therapeutics industry, which can be attributed to a high asthma prevalence rate of 7.7% in the adult population. In addition to that, a well-developed reimbursement system exists in both private and public sectors to cover branded biologics. The high level of penetration of dupilumab, mepolizumab, benralizumab, and tezepelumab in the United States formulary system, along with a high level of direct-to-consumer pharmaceutical marketing campaigns and expanding patient support services, continues to fuel premium-priced biologic therapy in large numbers in hospital, outpatient, and home care settings.
Asthma Therapeutics Market Growth Drivers:
-
Rising Global Asthma Prevalence and Unmet Treatment Need Are Fueling Sustained Asthma Therapeutics Market Growth
The rise in global cases of asthma, fueled by factors such as urbanization, deterioration in air quality, sedentary lifestyles, and increased exposure to occupational allergens, is still the primary factor driving the market for asthma therapeutics in terms of market size. With more than 339 million people worldwide estimated to be living with asthma as of 2025, the existing gap in terms of optimal treatment attainment is still a key factor driving the market for asthma therapeutics. According to the CDC, more than 1.6 million emergency department visits in the United States are attributed to asthma each year, further adding to the compounded need for better systems of managing this condition in the future. The rise in cases of asthma-COPD overlap syndrome is also adding to the market for asthma therapeutics, as this population requires dual-indication treatment regimens.
For instance, in October 2024, AstraZeneca reported that its respiratory biologics portfolio, including Fasenra (benralizumab) and Breztri Aerosphere (budesonide/glycopyrrolate/formoterol), generated combined global sales exceeding USD 3.2 billion for the fiscal year, reflecting accelerating biologic penetration within the severe asthma treatment landscape.
Asthma Therapeutics Market Restraints:
-
High Cost of Biologic Therapies and Formulary Access Barriers Are Hampering Asthma Therapeutics Market Penetration
While the clinical effectiveness of these next-generation biologic agents in the treatment of asthma has been well proven, the prohibitively high cost of treatment with these agents in the United States, which ranges between USD 15,000 to over USD 40,000 annually per patient, remains a major obstacle to wider adoption of these agents in the treatment of asthma in low- and middle-income economies where out-of-pocket payments are a major determinant of healthcare expenditures. In addition to these issues, stringent prior authorization requirements in many health insurance plans in the United States, along with step therapy requirements that mandate failure of traditional ICS/LABA therapy prior to access to biologic agents, have also limited access to these agents. In addition, a lack of biosimilars to many of the well-established biologic agents has also been a major factor in limiting access to these agents in many emerging economies in Southeast Asia, Sub-Saharan Africa, and Latin America due to a lack of awareness among primary care physicians in these economies regarding advanced phenotyping in asthma treatment.
Asthma Therapeutics Market Opportunities:
-
Biologic Therapy Pipeline Expansion and Biosimilar Entry Create High-Value Growth Opportunities for the Asthma Therapeutics Market
The strong pipeline of novel biologic agents targeting new emerging asthma endotypes such as thymic stromal lymphopoietin (TSLP), IL-33, and prostaglandin D2 receptor (DP2) represents transformative business growth opportunities for both originator pharmaceutical and specialty biologic manufacturers. The approval of tezepelumab as a first biologic effective in broad-spectrum asthma phenotypes regardless of eosinophil count has expanded the patient population for biologic therapy and created a template for broad-spectrum respiratory immunology therapies. Meanwhile, the future launch of biosimilar omalizumab, mepolizumab, and benralizumab between 2026 and 2030 is expected to make access more democratic, minimize payer pushback, and expand the total available market by adding cost-prohibitive patient segments in North America, Europe, and Asia Pacific.
For instance, in January 2025, the FDA approved Nucala (mepolizumab) biosimilar applications from two manufacturers, with commercial launches anticipated in mid-2025, expected to reduce annual biologic treatment costs by 25–40% and substantially broaden patient access within U.S. managed care networks.
Asthma Therapeutics Market Segment Analysis
-
By product, inhalers held the dominant revenue share of approximately 72.14% in 2025, while nebulizers are expected to register steady growth driven by pediatric and elderly patient populations.
-
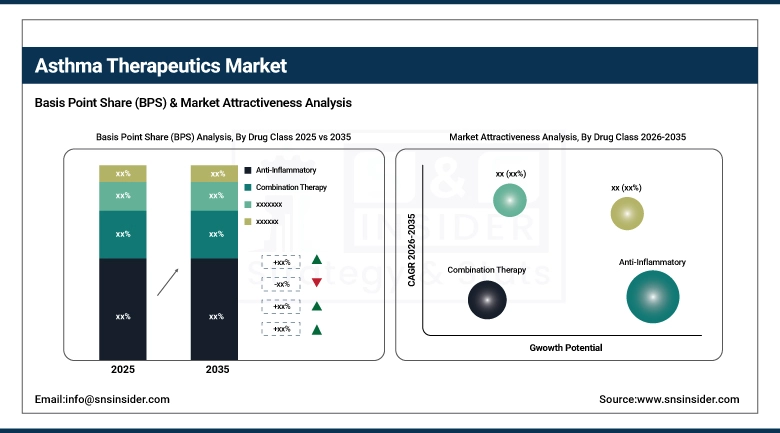
By drug class, anti-inflammatory agents accounted for the largest market share of around 44.86% in 2025, and combination therapy is projected to grow at the highest CAGR of 6.21% through 2035.
-
By route of administration, the inhaled segment led the market with approximately 68.43% share in 2025, while the oral segment is expected to expand with the rising adoption of oral biologics and leukotriene receptor antagonists.
By Drug Class, Anti-Inflammatory Agents Dominate While Combination Therapy Registers Fastest Growth
Anti-inflammatory agents, which include inhaled corticosteroids (ICS) like fluticasone propionate, budesonide, and beclomethasone dipropionate, held the highest drug class revenue share of 44.86% of the overall asthma therapy market in 2025. This is due to the fact that anti-inflammatory agents are recommended by all guidelines for the long-term management of asthma. Moreover, the wide acceptance of anti-inflammatory agents by all severity levels of asthma and the presence of generic ICS products at affordable prices have contributed to the dominance of the anti-inflammatory segment. The combination therapy segment of ICS/LABA and ICS/LABA/LAMA fixed-dose combinations is expected to grow at the highest compound annual growth rate of 6.21% from 2025 to 2035. This is due to the increasing clinical evidence of the benefits of using triple therapy once daily for the management of moderate-to-severe asthma to prevent severe exacerbations of the disease. Bronchodilators like SABAs and LABAs are essential drugs for the management of asthma. They have been maintaining stable market relevance across all geographic segments.
By Product, Inhalers Lead the Market While Nebulizers Register Consistent Growth
The segment of inhalers accounted for the largest share of the asthma therapy market in 2025, accounting for 72.14% of the total revenue. This is due to the strong clinical preference for direct airway drug delivery, the presence of well-established prescribing guidelines that endorse the use of ICS-containing inhalers as first-line controller therapy for asthma, and the innovation pipeline of DPI, MDI, and SMI devices. In the segment of inhalers, the share of MDIs is the highest among the devices. MDIs are supported by their efficacy, cost-effectiveness, and familiarity among prescribers. DPIs are also increasing due to the propellant-free formulation of the drug. In the segment of inhalers, the share of SMI devices is also increasing due to the low force of inhalation required by the elderly and pediatric patients. The segment of nebulizers is the smallest contributor to the revenue of the asthma therapy market. However, the segment is expected to grow steadily due to the high usage of nebulizers in the hospital setting, intensive care units, and home care programs for patients experiencing severe or near-fatal asthma exacerbations.
By Route of Administration, Inhaled Segment Leads While Oral Segment Gains Momentum
The route of administration of inhalation retained its dominant position at approximately 68.43% revenue share in 2025. This is because clinical guidelines have universally recommended the inhalation route of administration of both controller and reliever asthma medication due to the direct deposition of the drug into the airways. Moreover, the onset of action of the drug is quicker, and the systemic side effect profile is beneficial. In addition, the technology of inhaler devices is improving the bioavailability of the drug. The segment of the oral route of administration is picking up pace due to the increasing use of leukotriene receptor antagonists like montelukast. In addition, the use of systemic corticosteroids is increasing in the treatment of acute severe exacerbations of asthma. The segment of the “others” category is expanding rapidly due to the increasing use of injectable biologics like anti-IL-5 monoclonal antibodies and subcutaneous route of administration of monoclonal antibodies like anti-IgE.
Asthma Therapeutics Market Regional Highlights:
North America Asthma Therapeutics Market Insights:
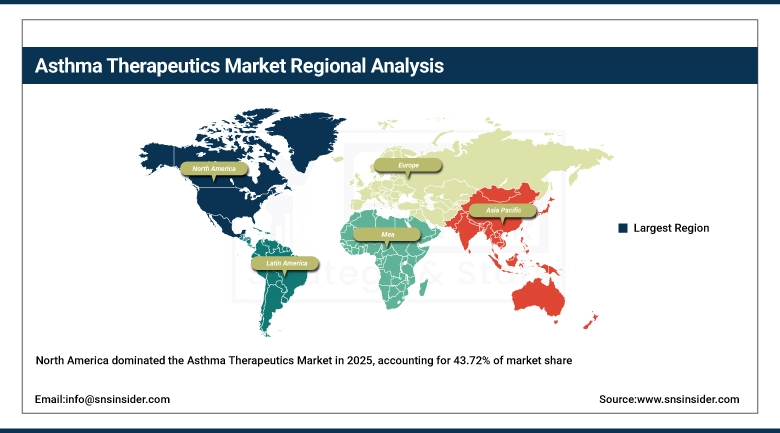
Over 43.72 % of regional revenue share in 2025, was held by North America, owing to high rates of asthma prevalence in the U.S., high percentage of key pharmaceutical innovators, established specialist respiratory care systems, and solid national reimbursement coverage for high-value biologic therapies. This is adding a significant portion to the regional market size in Canada as it is expanding reimbursement indications in Canada for its public drug program for biologic therapies. The eventual U.S. market is getting a boost from expedited FDA approval processes with regards to Breakthrough Therapy designation with respect to more severe cases of asthma, while high adoption of digital health monitoring solutions including connected inhalers and telemedicine based solutions for management of asthma are improving patient compliance and reducing preventable hospitalizations, thus improving market returns for players in the industry.
Get Customized Report as per Your Business Requirement - Enquiry Now
Asia Pacific Asthma Therapeutics Market Insights:
Asia Pacific is the fastest-growing market for asthma therapeutics, and it is expected to grow at a CAGR of 6.84% during the period of 2026 to 2035. This is due to the increasing rates of asthma in the region, especially in populated cities. In addition, the region is witnessing an increase in the purchasing power of residents, enabling them to access medical care. Furthermore, the government is increasing investment in health programs. China, India, Japan, and South Korea are the largest prescription markets in the region. China is increasingly incorporating advanced controller medications in its National Reimbursement Drug List. India is benefiting from the country's generic pharmaceutical manufacturing strength, enabling affordable access to inhalers and bronchodilators in the country's rural and semi-urban patient populations. In addition, Japan and South Korea are witnessing an increase in their elderly patient pool, enabling the use of advanced inhaler technologies and biologics for the growing segment of elderly-onset severe asthma.
Europe Asthma Therapeutics Market Insights:
Europe is home to the second-largest asthma therapeutics market in the world, fueled by universal access to care in major economies in Europe, well-developed regulatory systems in Europe through the EMA, and growing physician adoption of GINA Step Up Therapy approaches that include biologics in the treatment of severe uncontrolled asthma. The UK's NICE technology appraisal process has added to the label of mepolizumab, benralizumab, and dupilumab in asthma, whereas France, Germany, and Italy are launching national asthma plans that focus on specialist referral and biologic therapy screening in asthma. Europe's drive towards a carbon-neutral, HFC-free inhaler platform is also fueling R&D investment in low-carbon MDI propellants and dry powder devices.
Latin America (LATAM) and Middle East & Africa (MEA) Asthma Therapeutics Market Insights:
Latin America and Middle East & Africa are considered to be emerging opportunity zones in the asthma therapeutics industry, given the increase in urban air pollution levels, the rate of allergic sensitization in populations, and government initiatives to enhance surveillance and treatment of respiratory diseases. In LATAM, Brazil and Mexico are considered to be major contributors to the brand inhaler market and biologic industry growth. In Middle East & Africa, the Gulf Cooperation Council (GCC) nations are heavily investing in hospital formulary expansions and national respiratory care guidelines. Affordable generic inhaler availability, along with patient education campaigns in various languages and public-private partnership initiatives in the healthcare industry, are increasingly contributing to the diagnosis rate of asthma in both LATAM and Middle East & Africa, which are likely to act as a commercial base for future growth.
Asthma Therapeutics Market Competitive Landscape:
AstraZeneca PLC, established in 1999, is a global leader in respiratory therapy through its use of a diverse portfolio of biologic agents, fixed-dose combination inhalers, and "next-generation" precision medicines for patients with severe asthma. It has positioned itself at the forefront of severe asthma biologic therapy through its commitment to science-led innovation and extensive phase III programs, along with its commercial partnerships.
-
In March 2025, AstraZeneca received expanded FDA approval for Fasenra (benralizumab) as an add-on maintenance therapy for eosinophilic asthma patients aged 6 to 11 years, extending its biologic reach into the pediatric severe asthma segment and reinforcing its market leadership in age-inclusive biologic treatment.
GlaxoSmithKline plc / GSK (founded in 2000) is a global biopharmaceutical company with a dedicated respiratory franchise based on Nucala (mepolizumab), Arnuity Ellipta (fluticasone furoate), and the Ellipta delivery device. GSK continues to invest in the biology of eosinophils, biomarker-driven prescription, and value-based agreements with payers to drive biologic therapy in severe asthma.
-
In November 2024, GSK and Innoviva announced positive phase IIb data for an investigational once-daily oral CRTH2 antagonist for mild-to-moderate asthma, signaling the company's strategic expansion beyond injectable biologics into convenient oral precision respiratory therapy.
Sanofi S.A. (est. 1973), in partnership with Regeneron Pharmaceuticals, markets Dupixent (dupilumab), the first biologic targeting the IL-4/IL-13 pathway approved for moderate-to-severe asthma. Dupixent's unique mechanism, multi-indication profile, and demonstrated efficacy irrespective of eosinophil count have established it as one of the fastest-growing respiratory biologic franchises globally.
-
In January 2025, Sanofi and Regeneron reported that Dupixent global net sales exceeded USD 14.2 billion across all approved indications in 2024, with the asthma indication representing one of the highest-growth contributory segments, supported by expanded pediatric approval and broadening payer coverage in Europe and Asia Pacific.
Asthma Therapeutics Market Key Players:
-
AstraZeneca PLC
-
GlaxoSmithKline plc (GSK)
-
Sanofi S.A.
-
Regeneron Pharmaceuticals, Inc.
-
Boehringer Ingelheim GmbH
-
Novartis AG
-
Teva Pharmaceutical Industries Ltd.
-
Merck & Co., Inc. (MSD)
-
Pfizer Inc.
-
Roche Holdings AG (Genentech)
-
Amgen Inc.
-
Johnson & Johnson (Janssen Pharmaceuticals)
-
Chiesi Farmaceutici S.p.A.
-
Cipla Limited
-
Sun Pharmaceutical Industries Ltd.
-
Almirall S.A.
-
Vectura Group plc
-
Glenmark Pharmaceuticals Ltd.
-
Hikma Pharmaceuticals PLC
-
Theravance Biopharma, Inc.
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 29.58 Billion |
| Market Size by 2035 | USD 48.50 Billion |
| CAGR | CAGR of 5.07% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Product (Inhalers {Dry Powder, Metered Dose, Soft Mist}, and Nebulizers) • By Drug Class (Anti-inflammatory, Combination Therapy, and Bronchodilators) • By Route of Administration (Inhaled, Oral, and Others) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | AstraZeneca PLC, GlaxoSmithKline plc (GSK), Sanofi S.A., Regeneron Pharmaceuticals, Inc., Boehringer Ingelheim GmbH, Novartis AG, Teva Pharmaceutical Industries Ltd., Merck & Co., Inc. (MSD), Pfizer Inc., Roche Holdings AG (Genentech), Amgen Inc., Johnson & Johnson (Janssen Pharmaceuticals), Chiesi Farmaceutici S.p.A., Cipla Limited, Sun Pharmaceutical Industries Ltd., Almirall S.A., Vectura Group plc, Glenmark Pharmaceuticals Ltd., Hikma Pharmaceuticals PLC, Theravance Biopharma, Inc. |
Frequently Asked Questions
The Asthma Therapeutics Market is expected to reach USD 48.50 billion by 2035, indicating steady long-term growth across developed and emerging regions.
North America leads the Asthma Therapeutics Market with a 43.72% market share in 2025, supported by strong healthcare infrastructure and high biologic adoption.
The Asthma Therapeutics Market is projected to grow at a CAGR of 5.07% over the forecast period from 2026 to 2035.
In the Asthma Therapeutics Market, inhalers dominate with a 72.14% revenue share in 2025, driven by their clinical effectiveness and widespread adoption.
The Asthma Therapeutics Market was valued at USD 29.58 billion in 2025, reflecting strong demand driven by rising global asthma prevalence and expanding treatment options.

