Blood Transfusion Diagnostics Market Size Analysis:
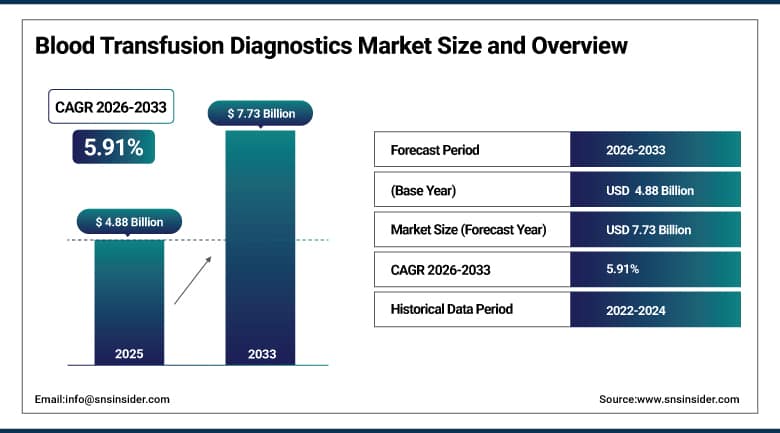
The Blood Transfusion Diagnostics Market was valued at USD 4.88 billion in 2025E and is expected to reach USD 7.73 billion by 2033, growing at a CAGR of 5.91% from 2026-2033.
The comprehensive analysis of the Blood Transfusion Diagnostics market includes market taxonomy and trends, key statistical insights, and the incidence and prevalence of blood disorders requiring transfusions in 2023. The report details prescription trends by region, device volume across regions, and healthcare spending by government, commercial, private, and out-of-pocket expenditures. Furthermore, it analyzes the increasing reliance on automated systems of diagnostics, the importance of updated blood screening technologies, and the rapidly growing demand for precision diagnostics in blood transfusions. It also discusses the trends in regulatory frameworks and their impact on diagnostic device adoption, providing a comprehensive view of the market growth.
Market Size and Forecast
-
Market Size in 2025E: USD 4.88 Billion
-
Market Size by 2033: USD 7.73 Billion
-
CAGR: 5.91% from 2026 to 2032
-
Base Year: 2025
-
Forecast Period: 2026–2033
-
Historical Data: 2022–2024
To Get More Information On Blood Transfusion Diagnostics Market - Request Free Sample Report
Blood Transfusion Diagnostics Market Trends
-
Rising prevalence of blood disorders and increasing demand for safe transfusions are driving the blood transfusion diagnostics market.
-
Growing adoption of automated testing, immunoassays, and molecular diagnostics is enhancing accuracy and efficiency.
-
Expansion of hospitals, blood banks, and healthcare infrastructure is boosting market growth.
-
Increasing focus on transfusion safety, donor screening, and regulatory compliance is shaping adoption trends.
-
Advancements in rapid testing kits and point-of-care diagnostics are improving accessibility and turnaround times.
-
Integration with digital health records and laboratory informatics is supporting workflow optimization.
-
Collaborations between diagnostic companies, healthcare providers, and research institutions are accelerating innovation and technology adoption.
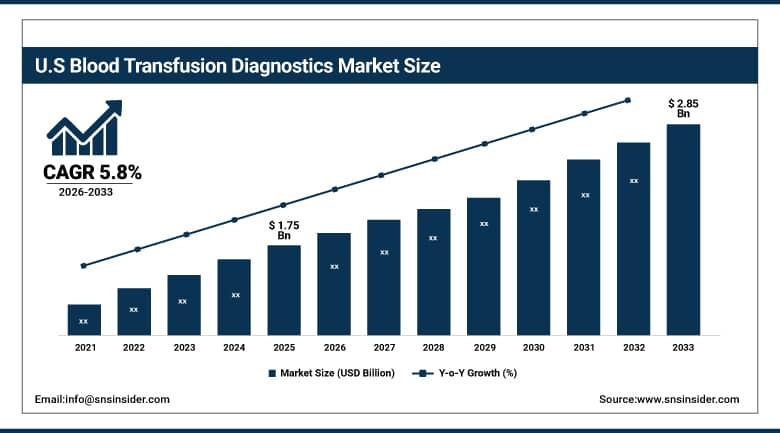
The U.S. Blood Transfusion Diagnostics Market size was valued at an estimated USD 1.75 billion in 2025 and is projected to reach USD 2.85 billion by 2033, growing at a CAGR of 5.8% over the forecast period 2026–2033. Market growth is driven by the increasing demand for safe blood transfusions, rising prevalence of chronic diseases, and growing number of surgical procedures and trauma cases across the country. Advancements in immunohematology and molecular diagnostic technologies, along with stringent regulatory requirements for blood screening and compatibility testing, are supporting market expansion. Additionally, strong healthcare infrastructure, widespread adoption of automated diagnostic systems, and continuous innovation by leading diagnostic manufacturers further contribute to the steady growth of the U.S. blood transfusion diagnostics market during the forecast period.
Blood Transfusion Diagnostics Market Growth Drivers:
-
Rising Incidence of Blood Disorders and Hematological Diseases propelling the Blood Transfusion Diagnostics Market growth.
Blood-related disorders, such as anemia, thalassemia, hemophilia, and sickle cell disease, are all on the rise, and coupling that with the out-of-control blood boom around the globe is one of the most powerful inducers for the Blood Transfusion Diagnostics market. As per the data from the World Health Organization (WHO), almost 1.62 billion globally suffer from anemia, with high rates being reported in countries such as sub-Saharan Africa and South Asia. This increasing prevalence fuels the demand for effective and accurate diagnostics for blood screening and transfusion. The rising number of surgeries and trauma cases needing blood transfusion is also fueling the demand for diagnostic tools to ensure blood type compatibility and reduce transfusion reactions, thus boosting the global market.
-
Advancements in Blood Transfusion Diagnostic Technology accelerating the market growth
Technological advancements in diagnostics for blood transfusion, especially in blood screening and blood-typing systems, are driving the growth of the market. The emergence of next-generation innovative, AI-enabled diagnostic tools and high-throughput blood screening devices. The data for May 2024, for instance, Thermo Fisher Scientific launched the Applied Biosystems Axiom BloodGenomiX Array, which provides a high degree of accuracy in the detection of extended blood groups and rare blood groups that can help in the optimization of blood transfusion matching. These advancements help enhance the accuracy of blood transfusion diagnostics that minimize transfusion-related complications and, thereby, increase the demand for advanced systems at clinical as well as emergency sites.
Blood Transfusion Diagnostics Market Restraints:
-
High Cost of Diagnostic Systems Restraining the Blood Transfusion Diagnostics Market Growth.
The high expenditure involved in advanced blood transfusion diagnostic systems is one of the major restraining factors that are expected to hinder the market during the forecast period, which in turn is anticipated to impede market growth, particularly in low- and middle-income regions. Investing heavily in infrastructure and training for the solutions of state-of-the-art technology, such as high-throughput blood screening and artificial intelligence diagnostic tools This may result in low adoption, particularly in resource-poor environments, where budgetary constraints once again limit access to such advanced solutions. Consequently, the cost factor limits their widespread applicability in several hospitals and clinical laboratories, which hampers their market growth in various regions. Moreover, these systems require continual upkeep and maintenance, which entails further costs that make them unreachable for healthcare facilities with rattled budgets.
Blood Transfusion Diagnostics Market Opportunities:
-
The integration of artificial intelligence (AI) and automation in blood transfusion diagnostics presents a significant market opportunity.
Diagnosis AI can bring about unprecedented accuracy, speed, and efficiency in screening for infections and matching blood types. Automation can reduce the possibility of human error and lower labor costs, and it is attractive to healthcare providers looking to improve operational efficiency and patient outcomes. With healthcare systems across the globe moving towards the adoption of digital technologies to improve diagnostic procedures, the demand for AI-based solutions in blood transfusion diagnostics is forecasted to increase. This trend opens great growth opportunities for companies that are designing new-generation diagnostic tools and systems based on such innovative technologies.
Blood Transfusion Diagnostics Market Challenges:
-
The blood transfusion diagnostics market faces significant challenges related to stringent regulatory requirements and compliance standards.
Stringent regulatory requirements and compliance standards are among the key obstacles hindering the growth of the blood-transfusion diagnostics market. Manufacturers can face challenges via disparate regulatory practices around the world, resulting in difficulties in achieving harmonized access to important markets for diagnostic products. New diagnostic technologies face numerous regulatory hurdles with extensive approval processes that can delay the launch of products or inhibit them altogether from entering the market. Further expansion of regulatory policies, especially in regions with intricate healthcare procedures, may pose challenges for compliance, further increasing the risk of product recall, payment of fines, or delay in market launch. The pile-up of multi-country regulations numbers and the overall complexity pose challenges to companies that intend to bring forth innovative solutions in the blood transfusion diagnostics landscape.
Blood Transfusion Diagnostics Market Segment Analysis
By Product, Reagents & Kits dominate with 65.23% market share, driven by critical role in accurate blood testing and ongoing innovations.
The Reagents & Kits segment dominated the market and accounted for the 65.23% market share in 2025 of the Blood Transfusion Diagnostics Market due to their critical role in ensuring accurate and reliable blood testing procedures. The reagents and kits are used for blood typing, infectious disease screening, and cross-matching, which are interdependent and important steps in the blood transfusion process. Their increasing demand is due to the ever-expanding need for effective and safe blood transfusion, especially in hospitals, blood banks, and diagnostic laboratories. Their ubiquity is also boosted by pre-packaged kits for use and standardized testing. Never-ending innovations in this segment, including better reagents for rare blood types and improved kits for quick results, are still maintaining the dominance of this segment in the market.
Ongoing improvements in output precision and test materials sensitivity are expected to be a key driver of growth of this segment, especially on account of rising patient safety and quality during blood transfusion processes. As the standards and regulations in blood testing increase, the demand for high-quality, reliable reagents and kits is also increasing rapidly, solidifying this segment's market leadership. With the increase in blood donation programs around the world, the demand for such diagnostic products will continue to rise, thereby propelling growth.
By Application, Disease Screening leads with 62.32% share, fueled by emphasis on infectious disease detection and global blood safety initiatives.
The Disease Screening segment dominated the market with 62.32% market share in 2025 the Blood Transfusion Diagnostics Market is also projected to register the fastest growth throughout the forecast period as an increasing emphasis is being put on screening blood for infectious diseases consisting of HIV, Hepatitis, and other infectious microorganisms. The widespread implementation of disease screening technologies has been mainly driven by a greater focus on blood safety. Moreover, the increasing global health initiatives to reduce the transmission of several infectious diseases via blood transfusion are also stimulating the significant demand for disease screening tools.
The blood screening instruments and analyzers segment is owing to the increasing awareness regarding blood safety, as well as the rising number of blood transfusions every year. Technology has also been developed to create new diagnostic tools, improve real-time screening tests, and use automated systems to enhance the efficiency and accuracy of disease screening, making it easier to integrate the latest tests into the blood transfusion process. Additionally, an increasing number of blood donors coupled with the growing incidence of chronic diseases are propelling the market's swift expansion.
By End-Use, Blood Banks hold 40.25% market share, with fastest growth expected due to rising blood transfusion demand and advanced screening technologies.
The blood banks segment dominated the market with 40.25% of the market share in 2025. Blood banks are integral points for blood collection, storage, and testing, which needs high-accuracy diagnostic tools for detecting infectious diseases, blood types, and other essential facets. As awareness about blood safety, as well as stringent regulations, have increased, blood banks have become vital in ensuring that only safe blood is provided to the patients. This has propelled the demand for preventive diagnostic instruments, thereby establishing the segment's expertise in the market.
This segment is likely to witness the fastest growth during the forecast period owing to rising global demand for blood products, especially in emerging economies with growing healthcare infrastructure. The growing number of chronic diseases like cancer and anemia that require regular transfusion and the increasing frequency of trauma and surgical procedures also fuel the demand for blood banks. Moreover, the growth in this segment can also be attributed to advancements in blood screening technology and government initiatives for safe blood donation and transfusion.
Blood Transfusion Diagnostics Market Regional Analysis
North America Blood Transfusion Diagnostics Market Insights
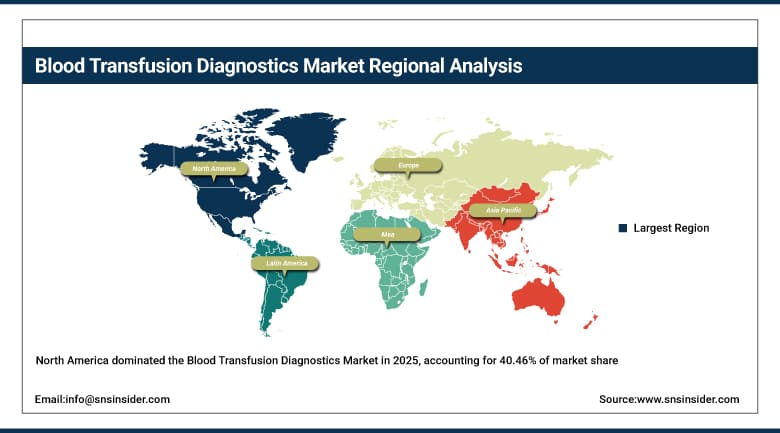
North America dominated the market with 40.46% of the market share in 2025 due to advanced healthcare infrastructure, widely adopted innovative diagnostic technologies, and well-established healthcare policies in the region contributing to its market share. The rapid availability of advanced blood transfusion diagnostic tools is attributed to the presence of market leaders, including Thermo Fisher Scientific Illumina, among others. In addition, the high incidence of blood-related diseases, in conjunction with the widespread government and private sector investments in healthcare, also supports the growth of the blood transfusion diagnostics market. The North American regulatory framework further reinforces this, providing stringent quality controls for diagnostics, leading to faster approvals and greater market share.
Get Customized Report as Per Your Business Requirement - Enquiry Now
Asia Pacific Blood Transfusion Diagnostics Market Insights
The blood transfusion diagnostics market in Asia Pacific is predicted to grow the fastest at a CAGR of 8.62% in the forecasted period 2026-2033, owing to the speedy development of healthcare infrastructure, growing medical tourism, and rising government expenditure on healthcare services. Countries such as China and India are making great strides in improving their healthcare systems and access to advanced diagnostics, including blood transfusion testing. Moreover, the increasing occurrence of blood disorders and the geriatric population in the regions further propel the demand for precise and efficient diagnostics. Owing to rising awareness and affordability of health care, especially with more cost-effective diagnostic solutions entering the market.
Europe Blood Transfusion Diagnostics Market Insights
The Europe Blood Transfusion Diagnostics Market is expanding due to rising demand for safe blood supply, increasing prevalence of blood-related disorders, and stringent regulatory standards. Advanced diagnostic technologies, including automated blood typing, pathogen detection, and nucleic acid testing, are enhancing accuracy and efficiency. Growing awareness of transfusion safety, adoption of innovative assays, and increasing investments in healthcare infrastructure across countries like Germany, France, and the UK are driving market growth throughout the region.
Middle East & Africa and Latin America Blood Transfusion Diagnostics Market Insights
The Blood Transfusion Diagnostics Market in the Middle East & Africa and Latin America is growing due to increasing demand for safe blood supply, rising prevalence of blood disorders, and healthcare infrastructure development. Adoption of advanced diagnostic technologies, government initiatives for transfusion safety, and awareness programs are driving growth. Expansion of hospitals, blood banks, and laboratories, along with collaborations between diagnostic providers and healthcare organizations, further supports market development across these regions.
Blood Transfusion Diagnostics Market Competitive Landscape:
Siemens Healthineers
Siemens Healthineers, founded in 1847 and headquartered in Erlangen, Germany, is a global leader in medical technology, providing diagnostic imaging, laboratory diagnostics, and advanced therapy solutions. The company focuses on innovation in imaging, molecular diagnostics, and digital health to enhance patient care and operational efficiency. Siemens Healthineers delivers solutions for hospitals and clinics worldwide, including advanced radiopharmaceuticals, imaging systems, and integrated healthcare technologies that improve diagnosis, treatment, and patient outcomes.
-
December 10, 2024: Siemens Healthineers completed the acquisition of Advanced Accelerator Applications Molecular Imaging from Novartis. The Europe-based network, specializing in diagnostic radiopharmaceuticals for PET scans, will operate as Advanced Accelerator Applications under Siemens Healthineers.
Illumina, Inc.
Illumina, founded in 1998 and headquartered in San Diego, California, is a global leader in genomics, offering DNA sequencing and array-based solutions for research, diagnostics, and clinical applications. The company enables precision medicine and genomic research through innovative tools that profile genetic information, identify biomarkers, and support targeted therapies. Illumina’s technologies empower clinicians and researchers to improve patient outcomes, advance personalized medicine, and accelerate discoveries in oncology, immunology, and other therapeutic areas.
-
August 2024: The U.S. FDA approved Illumina’s TruSight Oncology (TSO) Comprehensive test as an in vitro diagnostic device. The test analyzes over 500 genes from solid tumors to profile patients’ tumors, enabling targeted therapy selection and participation in clinical trials for new drugs.
Thermo Fisher Scientific Inc.
Thermo Fisher Scientific, founded in 1956 and headquartered in Waltham, Massachusetts, is a leading global provider of analytical instruments, laboratory equipment, and life sciences solutions. The company focuses on advancing research, diagnostics, and clinical testing by offering innovative tools and high-throughput technologies. Thermo Fisher supports genomic, proteomic, and cellular analysis, enabling improved diagnostics, personalized medicine, and enhanced biomedical research across academic, industrial, and clinical applications worldwide.
-
May 2024: Thermo Fisher Scientific released the Applied Biosystems Axiom BloodGenomiX Array and software, enabling detection of complex and rare blood groups, HLA tissue types, and platelet types in a single high-throughput assay, improving donor blood matching and supporting innovation in transfusion medicine.
Blood Transfusion Diagnostics Market Key Players
-
Illumina, Inc. (Verifi Prenatal Test, TruSight NIPT Solution)
-
Thermo Fisher Scientific Inc. (Thermo Scientific Dako Blood Transfusion Kits, Thermo Scientific Invitrogen Immunoassays)
-
Abbott Laboratories (Alinity's System, PRISM HIV O Plus Assay)
-
Grifols (Procleix Panther System, Grifols Blood Typing Reagents)
-
Siemens Healthineers (ADVIA Centaur XP Immunoassay System, Atellica IM TSH)
-
Roche Diagnostics (Cobas e 411 Analyzer, Roche Diagnostics Blood Transfusion Kits)
-
Beckman Coulter, Inc. (Access 2 Immunoassay System, Coulter LH 750 Hematology Analyzer)
-
Becton Dickinson and Company (BD Max System, BD Vacutainer Blood Collection Tubes)
-
Ortho Clinical Diagnostics (Ortho Vision Analyzer, Ortho Workstation)
-
Bio-Rad Laboratories, Inc. (BioPlex 2200, Bio-Rad Transfusion Diagnostic Systems)
-
Akers Biosciences (The BreathScan Alcohol Breathalyzer, The Blood Typing Test)
-
MacoPharma (Macotest Blood Typing, MacoBlood IV-Drug Test)
-
F. Hoffmann-La Roche AG (Cobas 8000 Series, Roche Cobas Liat System)
-
Sysmex Corporation (Sysmex XN-Series, Sysmex XN-31 Hematology Analyzer)
-
Quidel Corporation (QuickVue Influenza A+B Test, QuickVue At-Home Test Kit)
-
Abcam (Human Blood Typing Reagents, Hemoglobin Subunit Antibodies)
-
BioMerieux (VIDAS Blood Culture System, BioFire FilmArray Blood Culture Panel)
-
Sekisui Diagnostics (Sapphire Blood Typing, Immunoassay Panels)
-
Diacor (Diacor Blood Typing Kits, Diacor Immunoassay Systems)
-
Wako Pure Chemical Industries, Ltd. (Human Blood Coagulation Reagents, Blood Typing Reagents)
Suppliers
-
Thermo Fisher Scientific Inc.
-
Beckman Coulter, Inc.
-
Bio-Rad Laboratories, Inc.
-
Siemens Healthineers
-
Roche Diagnostics
-
Illumina, Inc.
-
Grifols
-
Abbott Laboratories
-
Ortho Clinical Diagnostics
-
Becton Dickinson and Company
| Report Attributes | Details |
|---|---|
| Market Size in 2025E | US$ 4.61 Billion |
| Market Size by 2033 | US$ 7.73 Billion |
| CAGR | CAGR of 5.91% From 2026 to 2033 |
| Base Year | 2025 |
| Forecast Period | 2026-2033 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Product (Reagents & Kits, Instruments, Others) • By Application (Blood Grouping, Disease Screening) • By End-Use (Hospitals, Blood Banks, Diagnostics Laboratories, Others) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | Illumina, Inc., Thermo Fisher Scientific Inc., Abbott Laboratories, Grifols, Siemens Healthineers, Roche Diagnostics, Beckman Coulter, Inc., Becton Dickinson and Company, Ortho Clinical Diagnostics, Bio-Rad Laboratories, Inc., Akers Biosciences, MacoPharma, F. Hoffmann-La Roche AG, Sysmex Corporation, Quidel Corporation, Abcam, BioMerieux, Sekisui Diagnostics, Diacor, Wako Pure Chemical Industries, Ltd., and other players. |
Frequently Asked Questions
Ans: North America dominated the market in 2025 with a 40.46% share, supported by advanced healthcare infrastructure, technological adoption, and stringent regulatory standards for transfusion safety.
Ans: Reagents & Kits dominated the market with 65.23% share, owing to their critical role in accurate blood testing, innovation, and widespread adoption across diagnostics applications.
Ans: Rising incidence of blood disorders, increasing demand for safe transfusions, and the adoption of advanced diagnostic technologies are the key factors driving market growth.
Ans: The Blood Transfusion Diagnostics Market size in 2025 was valued at USD 4.88 billion, reflecting increasing adoption of automated testing, immunoassays, and molecular diagnostics globally.
Ans: The Blood Transfusion Diagnostics Market is expected to grow at a CAGR of 5.91% from 2026 to 2033, driven by rising blood disorder prevalence and transfusion demand.

