Hunter Syndrome Treatment Market Report Scope & Overview:
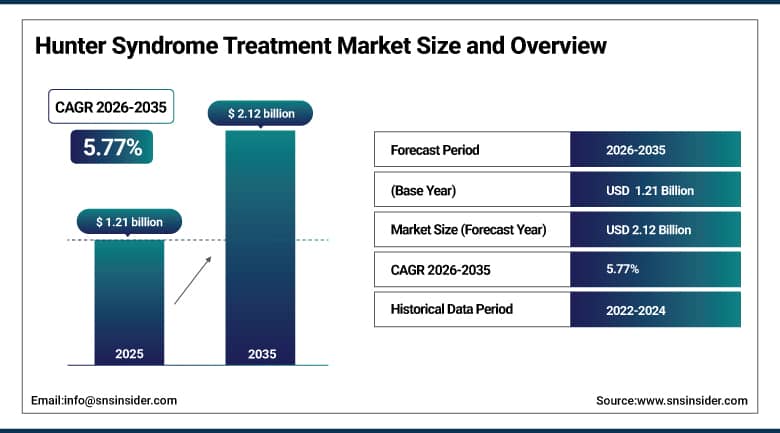
The Hunter Syndrome Treatment Market Size is valued at USD 1.21 Billion in 2025 and is projected to reach USD 2.12 Billion by 2035, growing at a CAGR of 5.77% during the forecast period 2026–2035.
Hunter Syndrome Treatment Market Analysis presents a brief description of the various treatment options available for treating this rare lysosomal storage disease. This report also highlights how the market will grow between 2026 and 2035. Growth will be attributed to increased diagnosis, improvement in treatments, and increased development of orphan drugs.
The Hunter Syndrome Treatment Market served over 10,000 patients in 2025, driven by improved diagnosis and expanding access to enzyme and gene therapies.
Market Size and Forecast:
-
Market Size in 2025: USD 1.21 Billion
-
Market Size by 2035: USD 2.12 Billion
-
CAGR: 5.77% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get More Information On Hunter Syndrome Treatment Market - Request Free Sample Report
Hunter Syndrome Treatment Market Trends:
-
Rising focus on rare disease diagnosis and newborn genetic screening is improving early detection of Hunter Syndrome cases.
-
Increasing adoption of enzyme replacement therapy (ERT) remains the standard of care, driving consistent treatment demand.
-
Rapid growth in gene therapy and CNS-targeted therapies is reshaping the long-term treatment landscape.
-
Expanding clinical research in intrathecal and intracerebroventricular delivery is improving neurological outcome management.
-
Rising orphan drug approvals and regulatory incentives are accelerating pipeline development and commercialization.
-
Increasing patient access programs and specialty care networks are improving long-term disease management and survival outcomes.
U.S. Hunter Syndrome Treatment Market Insights:
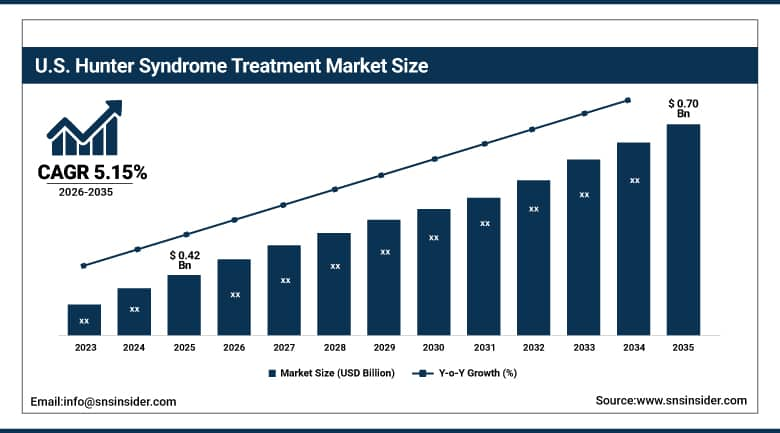
The U.S. Hunter Syndrome Treatment Market is projected to grow from USD 0.42 Billion in 2025 to USD 0.70 Billion by 2035, at a CAGR of 5.15%. The development process will be driven by strong diagnostics of rare diseases, the adoption of enzyme replacement therapy, gene therapy experiments, good orphan drug reimbursements, and increasing availability of centers for specialty treatments.
Hunter Syndrome Treatment Market Growth Drivers:
-
Rising rare disease awareness, genetic screening, and enzyme therapy adoption drive Hunter Syndrome treatment demand growth.
Hunter Syndrome Treatment Market dynamics include increased awareness about rare diseases, improvements in neonatal screening tests, and the availability of ERT. With advancements in genetic testing and improved clinical diagnosis of the condition, there will be more diagnoses made. Routine administration of ERT and potential gene therapy options will contribute to the growth in the market. There are also improvements in terms of supportive treatments and specialized clinics that are helping manage the condition better and improve patient outcomes.
Over 70% of diagnosed Hunter Syndrome patients in 2025 were pediatric cases, highlighting the strong role of early-onset genetic disorders in treatment demand.
Hunter Syndrome Treatment Market Restraints:
-
High treatment cost, limited curative options, and lifelong therapy burden restrain market accessibility and expansion.
There are many obstacles faced by the Hunter Syndrome Treatment Market, such as high expenses related to treatment, long-term nature of treatment procedures, and lack of effective solutions for curing the illness. The enzyme replacement therapy is still expensive and requires frequent administration; thus, it imposes considerable financial pressures on both patients and healthcare institutions. Additionally, being a rare disease, it complicates conducting extensive research on the subject matter and delays drug development processes.
Hunter Syndrome Treatment Market Opportunities:
-
Gene therapy advancements and CNS-targeted innovations create strong opportunities for curative Hunter Syndrome treatments.
There are emerging possibilities in the Hunter Syndrome treatment market due to innovations in gene therapy, drug delivery to the central nervous system, and modern biologics. Clinical studies conducted on enzyme delivery and gene editing can possibly change the face of treatment for Hunter Syndrome. An increase in orphan drugs is driving pharmaceutical companies to develop new treatments. With increased funding and personalized medicine, there could be better prognosis and even a possible cure for the disease.
Over 35% of pipeline therapies in 2025 focused on gene therapy and CNS-targeted delivery, reflecting the shift toward curative treatment innovation.
Hunter Syndrome Treatment Market Segmentation Analysis:
-
By Treatment Type, Enzyme Replacement Therapy (ERT) held the largest market share of 68.45% in 2025, while Gene Therapy is expected to grow at the fastest CAGR of 9.87% during 2026–2035.
-
By Route of Administration, Intravenous (IV) accounted for the highest market share of 71.28% in 2025, whereas Intrathecal / Intracerebroventricular (ICV) is projected to grow at the fastest CAGR of 8.95% during the forecast period.
-
By End User, Hospitals dominated with a 74.63% market share in 2025, while Specialty Clinics are expected to grow at the fastest CAGR of 8.21% through 2035.
-
By Patient Age Group, Pediatric segment held the largest share of 81.37% in 2025 and Adult patients are expected to grow at the fastest CAGR of 7.44% during 2026–2035.
-
By Disease Manifestation / Complication Type, Neurological complications dominated with a 29.54% share in 2025, while Musculoskeletal abnormalities are projected to grow at the fastest CAGR of 8.36% through 2035.
-
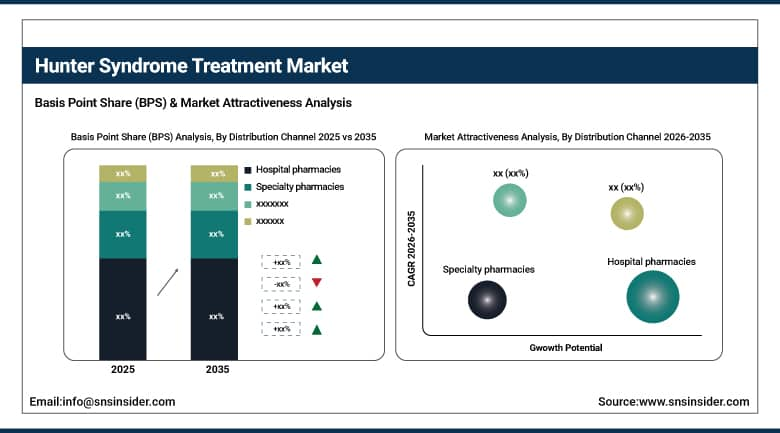
By Distribution Channel, Hospital Pharmacies held the largest share of 62.91% in 2025, whereas Specialty Pharmacies are expected to grow at the fastest CAGR of 8.73% during the forecast period.
By Distribution Channel, Hospital Pharmacies Dominated While Specialty Pharmacies Fastest Growing:
Hospital Pharmacies segment dominated the market owing to the centralization of dispensation of enzyme replacement therapies and their seamless integration with the hospital patient care infrastructure. By 2025, the total number of patients who had been administered these therapies via hospital pharmacies exceeded 7,500.
Specialty Pharmacies are the fastest growing segment driven by the need for care at home and chronic disease management. The year 2025 saw more than 3,000 patients using the services of specialty pharmacies owing to developments in cold chain distribution, better patient support, and the growth of rare disease networks.
By Treatment Type, Enzyme Replacement Therapy (ERT) Dominated While Gene Therapy Fastest Growing:
Enzyme Replacement Therapy (ERT) segment dominated the market owing to its already established practice within the healthcare domain as the gold standard treatment along with extensive availability in hospitals and specialized centers. Being provided regularly to the patients for treating their condition and improving their quality of life, the enzyme replacement therapy helped 7,000 patients in 2025.
Gene Therapy is the fastest growing segment owing to the increasing number of clinical trials, approval of more orphan drugs, and the high possibility of curing diseases through gene therapy. By 2025, an estimated 1,500 patients had been enrolled in the gene therapy program.
By Route of Administration, Intravenous (IV) Dominated While Intrathecal / Intracerebroventricular (ICV) Fastest Growing:
Intravenous (IV) segment dominated the market since it is still the major route for administering Enzyme Replacement Therapy. During 2025, over 8,000 patients were treated using this route, facilitated by existing hospital-based infusion facilities and widespread knowledge in the clinic.
Intrathecal / Intracerebroventricular (ICV) segment is the fastest growing segment due to an increasing focus on addressing the neurological issues that IV therapy cannot solve on its own. As of 2025, there were already more than 1,200 patients who had undergone CNS-specific drug delivery methods.
By End User, Hospitals Dominated While Specialty Clinics Fastest Growing:
Hospitals segment dominated the market on account of an excellent infrastructure required to handle disorders that were uncommon and needed infusions and treatment in a multidisciplinary manner. In 2025, the number of Hunter Syndrome patients treated in the hospitals reached 9,000+.
Specialty Clinics are the fastest growing segment due to the decentralization of rare diseases treatments, and the availability of outpatient care. In 2025, around 2,000 patients will have been diagnosed in the specialty clinics sector, owing to the increased awareness, and rare disease centers.
By Patient Age Group, Pediatric Dominated While Adult Patients Fastest Growing:
Pediatric segment dominated the market owing to the nature of the disease which primarily occurs during childhood due to its genetic origins. As of 2025, over 9,500 patients had been undergoing treatment, reflecting the importance of early diagnosis that results from newborn and genetic testing initiatives.
Adult is the fastest growing segment owing to survival rates and better management of the condition. It has been projected that there will be around 2,500 adults living in 2025, due to improvements in ERT and supportive care.
By Disease Manifestation, Neurological Complications Dominated While Musculoskeletal Abnormalities Fastest Growing:
Neurological complications segment dominated the market due to the high incidence rate and the impact on cognition and development among patients suffering from Hunter Syndrome. In the year 2025, over 6,000 incidences were reported with involvement of the neurological system.
Musculoskeletal abnormalities are the fastest growing segment due to an increased understanding of the importance of skeletal progression in long-term patients. The year 2025 saw about 3,000 cases diagnosed with musculoskeletal problems that were associated with their condition.
Hunter Syndrome Treatment Market Regional Analysis:
North America Hunter Syndrome Treatment Market Insights:
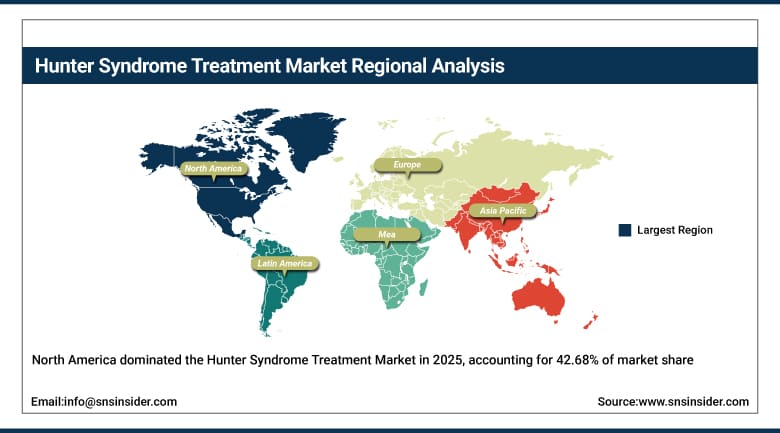
The market share held by the North America Hunter Syndrome Treatment Market, which makes up 42.68%, will see growth through its highly advanced rare disease health care system, genetic testing, and the use of enzyme replacement therapy. In North America, there is already a high number of hospitals, infusion centers, and excellent reimbursement for orphan drugs. Awareness levels for genetic diseases are high, and patients have access to early diagnosis and treatment. There are significant developments being made in gene therapy, precision medicine, and CNS-specific treatments.
Get Customized Report as Per Your Business Requirement - Enquiry Now
U.S. Hunter Syndrome Treatment Market Insights:
The U.S. Hunter Syndrome Treatment Market is spurred by extensive utilization of enzyme replacement therapy, strong newborn and genetic screening programs, and an efficient rare disease healthcare system. Key drivers include large biotech companies, the development of gene therapy through clinical studies, insurance, and special treatment facilities in the country.
Asia-Pacific Hunter Syndrome Treatment Market Insights:
The Asia-Pacific Hunter Syndrome Treatment Market is projected to grow at a CAGR of 7.90% during 2026–2035. The growth will be fueled by increased rare disease awareness, advancements in genetic testing programs, and increased diagnostic rates in China, India, Japan, and Southeast Asia. With growing healthcare facilities, availability of enzyme replacement treatments, and increasing use of gene therapy, the market is expected to experience consistent growth. Greater funding for rare disease studies, supportive government policies, and creation of rare disease treatment centers have helped accelerate patient diagnosis and treatment rates in the region.
China Hunter Syndrome Treatment Market Insights:
The China Hunter Syndrome Treatment Market is fueled by increasing awareness about rare diseases, developments in genetic screening techniques, and increased incidence rates. The development in the healthcare system, support from the government in the area of orphan disease study, availability of enzyme replacement treatment, and establishment of dedicated hospitals and biotech advancements have led to the growth of China Hunter Syndrome Treatment Market.
Europe Hunter Syndrome Treatment Market Insights:
The Europe Hunter Syndrome Treatment Market is driven by advanced health care facilities, high awareness of rare genetic diseases, and effective newborn screenings in Germany, France, and the United Kingdom. The diagnosis rate and accessibility of enzyme replacement treatment are driving factors that ensure stable demand. Orphan drugs, favorable reimbursement, and studies on genetic and brain-centered therapy are supporting the market as well. In addition, the rise in investments in specialized treatment clinics and cooperation of biotech companies and research institutes have positively impacted the European market.
Germany Hunter Syndrome Treatment Market Insights:
Germany is a key market in the Europe Hunter Syndrome Treatment Market considering its superior healthcare infrastructure, awareness regarding rare disorders, and its initiatives toward early detection through genetic testing. Rising numbers of diagnoses, increasing application of enzyme replacement therapies, favorable reimbursements, and research in gene therapy are some factors driving this market growth.
Latin America Hunter Syndrome Treatment Market Insights:
The Latin America Hunter Syndrome Treatment Market is witnessing growth owing to increased awareness regarding rare diseases and enhanced diagnostic methods for identifying such disorders through genetics in Brazil, Mexico, and Argentina. Increased availability of enzyme replacement treatments, improved healthcare facilities, and progressive establishment of treatment centers have contributed to market growth.
Middle East and Africa Hunter Syndrome Treatment Market Insights:
The Middle East and Africa Hunter Syndrome Treatment Market is growing driven by the advancement in healthcare facilities and increasing knowledge about genetic conditions. The increased rate of diagnoses, availability of enzyme replacement treatments, and the establishment of rare disease centers are contributing to the growth of the market. Countries such as Saudi Arabia, the UAE, and South Africa have embarked on rare diseases programs.
Hunter Syndrome Treatment Market Competitive Landscape:
Takeda Pharmaceutical Company Limited is one of the top biopharmaceutical companies and a major player in the Hunter Syndrome Treatment Market, mainly due to its enzyme replacement therapy (ERT) drug Elaprase (idursulfase). Takeda is supported by its strong distribution network, approval by regulators, and growing uptake by physicians. The company’s dominance will also get reinforced by its research work on rare diseases, extensive patient programs, and its attempt to improve market coverage in emerging countries.
-
In March 2025, Takeda expanded access to ELAPRASE (idursulfase), strengthening enzyme replacement therapy availability for Hunter Syndrome patients. Its rare disease commercialization initiatives reinforced its leadership in MPS II treatment and long-term patient support programs.
F. Hoffmann-La Roche Ltd plays an important role within the market for Hunter Syndrome Treatments due to its significant involvement in developing treatment methods for rare diseases and genetic conditions. It stands out among competitors due to its high-end biotechnological facilities, a strong pipeline of clinical research studies, and extensive knowledge about precision medicine and gene therapy. Roche enhances the diagnosis process by employing its highly developed diagnostics department. Investment into CNS-specific treatment methods, collaboration with other companies, and a broad global presence help maintain Roche’s position as an influential market leader.
-
In February 2025, Roche advanced its rare disease pipeline focusing on CNS-targeted biologics and improved delivery platforms. These efforts aim to enhance neurological treatment outcomes in lysosomal storage disorders, including Hunter Syndrome-related central nervous system complications.
Sanofi is one of the prominent international pharma firms with extensive participation in rare diseases management, particularly the provision of treatments in lysosomal storage disorders, which makes it an active participant in the Hunter Syndrome Treatment Market due to its expansive portfolio in biologics and specialty care. This is attributed to its strong research strength in enzyme therapies, wide presence across the globe, and focus on the development of orphan drugs. Its experience in rare metabolic conditions coupled with its partnerships and investments further consolidate its market position.
-
In April 2025, Sanofi progressed its rare disease pipeline through biologics and gene-based therapy expansion. The company strengthened its focus on enzyme and metabolic disorder innovations, supporting long-term treatment development for Hunter Syndrome and related lysosomal storage conditions.
Hunter Syndrome Treatment Market Key Players:
Some of the Hunter Syndrome Treatment Market Companies are:
-
Takeda Pharmaceutical Company Limited
-
F. Hoffmann-La Roche Ltd
-
Sanofi
-
BioMarin Pharmaceutical Inc.
-
Denali Therapeutics Inc.
-
REGENXBIO Inc.
-
Sangamo Therapeutics, Inc.
-
Ultragenyx Pharmaceutical Inc.
-
JCR Pharmaceuticals Co., Ltd.
-
ArmaGen Inc.
-
CANbridge Pharmaceuticals Inc.
-
Green Cross Corporation
-
Inventiva Pharma
-
Chiesi Farmaceutici S.p.A.
-
Orchard Therapeutics plc
-
uniQure N.V.
-
Freeline Therapeutics Holdings plc
-
Lysogene S.A.
-
Avrobio, Inc.
-
Regeneron Pharmaceuticals, Inc.
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 1.21 Billion |
| Market Size by 2035 | USD 2.12 Billion |
| CAGR | CAGR of 5.77% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Treatment Type (Enzyme Replacement Therapy (ERT), Hematopoietic Stem Cell Transplant (HSCT), Gene Therapy, Substrate Reduction Therapy, Supportive / Symptomatic Treatment) • By Route of Administration (Intravenous (IV), Intrathecal / Intracerebroventricular (ICV), Other Administration Routes) • By End User (Hospitals, Specialty Clinics, Diagnostic & Research Centers, Others) • By Patient Age Group (Pediatric, Adult Patients) • By Disease Manifestation / Complication Type (Neurological complications, Cardiovascular complications, Respiratory disorders, Musculoskeletal abnormalities, Ophthalmic & auditory complications, Gastrointestinal & others) • By Distribution Channel (Hospital Pharmacies, Specialty Pharmacies, Retail Pharmacies, Online Pharmacies) |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
| Company Profiles | Takeda Pharmaceutical Company Limited, F. Hoffmann-La Roche Ltd, Sanofi, BioMarin Pharmaceutical Inc., Denali Therapeutics Inc., REGENXBIO Inc., Sangamo Therapeutics, Inc., Ultragenyx Pharmaceutical Inc., JCR Pharmaceuticals Co., Ltd., ArmaGen Inc., CANbridge Pharmaceuticals Inc., Green Cross Corporation, Inventiva Pharma, Chiesi Farmaceutici S.p.A., Orchard Therapeutics plc, uniQure N.V., Freeline Therapeutics Holdings plc, Lysogene S.A., Avrobio, Inc., Regeneron Pharmaceuticals, Inc. |
Frequently Asked Questions
Ans: The Hunter Syndrome Treatment Market is projected to grow at a CAGR of 5.77% during the forecast period 2026–2035.
Ans: The market is valued at USD 1.21 Billion in 2025 and is expected to reach USD 2.12 Billion by 2035.
Ans: Growth is driven by rising rare disease awareness, increasing genetic screening programs, expanding enzyme replacement therapy adoption, and advancements in gene therapy and specialized care infrastructure.
Ans: By Treatment Type, Enzyme Replacement Therapy (ERT) dominated with a 68.45% share, while Gene Therapy is expected to grow at the fastest CAGR of 9.87% during 2026–2035.
Ans: North America led the market with a 42.68% share in 2025, while Asia-Pacific is the fastest-growing region, projected to expand at a CAGR of 7.90% during 2026–2035.

