Rapid Diagnostic Tests Market Report Scope & Overview:
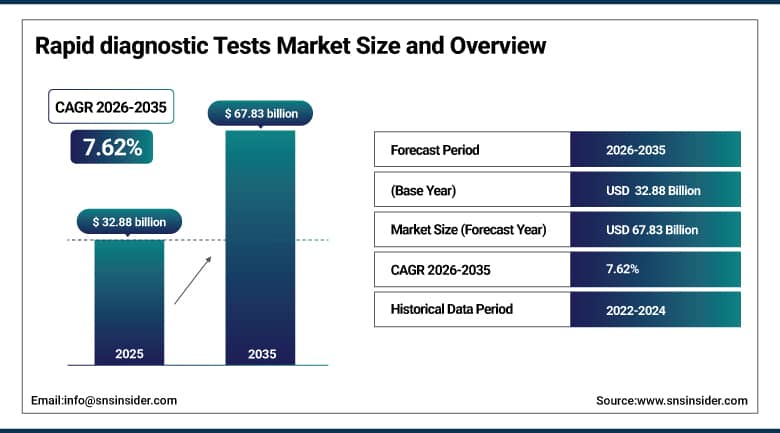
The Rapid Diagnostic Tests Market size is valued at USD 32.88 Billion in 2025 and is projected to reach USD 67.83 Billion by 2035, growing at a CAGR of 7.62% during the forecast period 2026–2035.
The Rapid Diagnostic Tests Market analysis report presents a comprehensive assessment of market dynamics, technological innovations, and diagnostic applications. The growing number of infections, the need for POCT, the use of home diagnostic tests, and improved healthcare infrastructure are expected to fuel market growth from 2026 to 2035.
There were more than 3.5 billion tests performed in 2025 due to increased screening programs, rapid and accurate diagnosis, and decentralization.
Market Size and Forecast:
-
Market Size in 2025: USD 32.88 Billion
-
Market Size by 2035: USD 67.83 Billion
-
CAGR: 7.62% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get More Information On Rapid diagnostic Tests Market - Request Free Sample Report
Rapid diagnostic Tests Market Trends:
-
Rising prevalence of infectious diseases such as COVID-19, influenza, dengue, and malaria is driving increased demand for rapid diagnostic tests.
-
Growing demand for point-of-care (POC) testing is boosting adoption of fast, accurate, and easy-to-use diagnostic solutions across healthcare settings.
-
Expansion of home-based and self-testing kits is improving accessibility, early diagnosis, and patient convenience.
-
Advancements in lateral flow assays, molecular diagnostics, and biosensor technologies are enhancing test accuracy, sensitivity, and speed.
-
Increasing integration of digital health tools and smartphone-enabled diagnostics is supporting real-time monitoring and data-driven decision-making.
-
Expansion of hospitals, diagnostic laboratories, and decentralized healthcare infrastructure is improving access to rapid diagnostic testing.
-
Government initiatives, large-scale screening programs, and public-private collaborations are accelerating market penetration and adoption.
U.S. Rapid diagnostic Tests Market Insights:
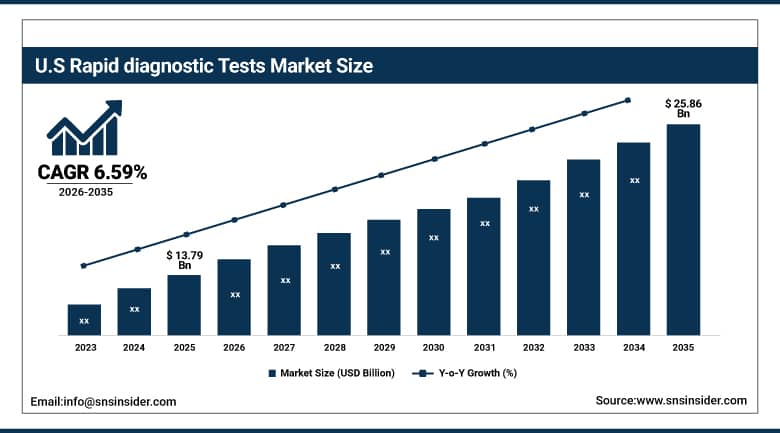
The U.S. Rapid Diagnostic Tests Market is projected to grow from USD 13.79 Billion in 2025 to USD 25.86 Billion by 2035, at a CAGR of 6.59%. The growth will be fueled by higher incidences of infections, higher demand for point of care diagnostics, extensive use of self-tests and home testing products, and the consistent development of rapid testing techniques in hospitals, clinics, and diagnostic laboratories.
Rapid diagnostic Tests Market Growth Drivers:
-
Rising prevalence of infectious diseases and increasing demand for rapid diagnosis driving adoption of advanced rapid diagnostic tests.
The increased incidence of infectious diseases such as COVID-19, influenza, dengue fever, and malaria, coupled with the requirement for early and reliable diagnosis, is driving the growth of the Rapid Diagnostic Test (RDT) Market. Hospitals, diagnostic centers, and home healthcare organizations are relying on point-of-care solutions, such as lateral flow immunoassays and molecular rapid tests, that provide quick decision-making capabilities. The development of portable, user-friendly, and high-sensitivity diagnostics, and the availability of self-diagnostic kits, is also aiding in the adoption process and driving the market forward.
More than 62% of hospitals, diagnostic centers, and homecare facilities are using RDTs for the detection of infectious diseases and screening applications in 2025.
Rapid diagnostic Tests Market Restraints:
-
Limited accuracy in certain conditions and regulatory challenges are restraining widespread adoption of rapid diagnostic tests in some healthcare settings.
The insensitivity and lack of specific tests of certain rapid diagnostic tests in cases of early-stage infections and low viral loads are critical barriers to the Rapid Diagnostic Tests Market. The potential for inconsistent results, together with possible errors of commission and omission, may undermine clinical assurance, requiring secondary laboratory tests, and thus, leading to increased medical expenditures. Regulatory hurdles, quality management challenges, and lengthy regulatory approval processes also pose obstacles to market penetration. Moreover, poor public recognition, insufficient capacity building in remote locations, and logistics difficulties in developing countries hamper widespread use, despite rising demand for rapid diagnostic methods.
Rapid diagnostic Tests Market Opportunities:
-
Expansion of home-based testing and digital-integrated diagnostics presents significant market growth opportunities.
As more focus has been put into decentralized healthcare and home care tests, tremendous market potential has emerged for the Rapid Diagnostic Tests Market. There is a rising adoption of self-testing kits and diagnostic products that provide immediate results without the need for laboratory testing facilities. The inclusion of rapid tests within mobile applications, telemedicine, and information exchange platforms makes it easier to monitor diseases, conduct early diagnosis, and manage patients remotely. Companies manufacturing consumer-oriented, cost-effective, and digitally integrated diagnostics can benefit from this trend, especially in infectious diseases and overall health monitoring.
In 2025, over 49% of the diagnostic testing was conducted with the help of rapid home or digitally-connected diagnostic testing kits.
Rapid Diagnostic Tests Market Segmentation Analysis:
-
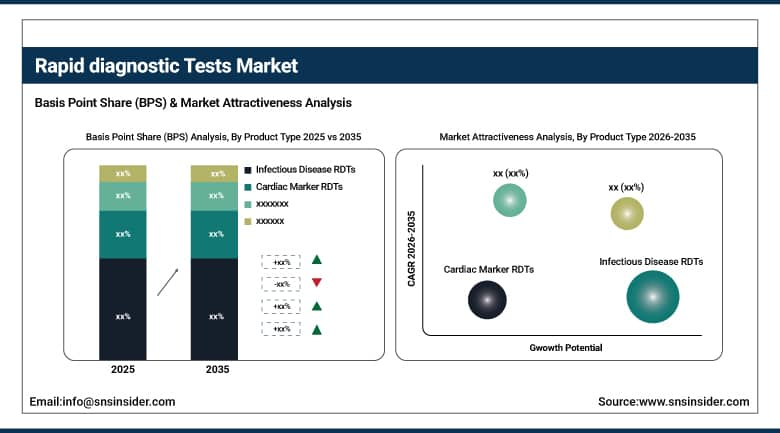
By Product Type, Infectious Disease RDTs held the largest market share of 52.75% in 2025, while Infectious Disease RDTs are also expected to grow at the fastest CAGR of 8.12% during 2026–2035.
-
By Technology, Lateral Flow Immunoassays (LFIA) dominated with 46.05% market share in 2025, whereas Molecular Rapid Tests are projected to record the fastest CAGR of 9.12% through 2026–2035.
-
By Application, Infectious Disease Testing accounted for the largest share of 48.85% in 2025, while Pregnancy & Fertility Testing is expected to grow at the fastest CAGR of 6.74% during the forecast period.
-
By End User, Hospitals & Clinics held the largest share of 45.12% in 2025, while Home Care Settings are expected to grow at the fastest CAGR of 8.54% during 2026–2035.
-
By Distribution Channel, Hospital Pharmacies dominated with 40.23% market share in 2025, whereas Online Pharmacies are projected to grow at the fastest CAGR of 9.47% through the forecast period.
By Product Type, Infectious Disease RDTs Dominate and Also Emerge as the Fastest-Growing Segment:
Infectious Disease RDTs segment dominated the market due to high demand for fast identification of infectious diseases along with large-scale screening activities in hospitals and laboratories. The high dependence on these diagnostics tests for accurate diagnosis and management of infectious disease outbreaks is expected to sustain their market leadership status in both developed and developing healthcare industries.
Infectious Disease RDTs are also the fastest-growing segment owing to an increasing incidence of infectious diseases around the world, coupled with screening programs launched by governments and consistent demand for fast diagnostics tools.
By Technology, Lateral Flow Immunoassays Dominate While Molecular Rapid Tests Grow Rapidly:
Lateral Flow Immunoassays segment dominated the market, being extensively employed due to its cost-effectiveness, user-friendliness, and fast processing time. The popularity of the product line in regular diagnostic procedures and mass screenings has ensured its ongoing use.
Molecular Rapid Tests are the fastest-growing segment, due to innovations in portable PCR technology and isothermal amplification techniques. Rising needs for more sensitive tests and accurate diagnosis have expedited the adoption of the test kits in hospitals and diagnostic centers.
By Application, Infectious Disease Testing Dominates While Pregnancy & Fertility Testing Grows Rapidly:
Infectious Disease Testing segment dominated the market due to the persistent burden of infectious diseases and the critical need due to the high prevalence of infectious diseases around the globe and the urgent requirement for rapid and accurate diagnosis at point-of-care or remote locations. Its continuous utilization in public health initiatives and emergencies has added strength to its position.
Pregnancy & Fertility Testing is the fastest-growing segment category, as it is witnessing increased awareness regarding early detection, preference for home diagnostic tests, and focus on reproductive health issues.
By End User, Hospitals & Clinics Dominate While Home Care Settings Grow Rapidly:
Hospitals & Clinics segment dominated the market due to these were the leading sites where patients went for quick diagnosis and clinical decision making. This segment continues to use point-of-care diagnostics due to they need rapid treatment for emergencies and infections along with routine screenings owing to the large number of patients.
Home Care Settings are the fastest-growing segment, pace due to growing adoption of home-based testing kits as people are demanding more accessible diagnostic technologies owing to the move toward decentralization of healthcare facilities.
By Distribution Channel, Hospital Pharmacies Dominate While Online Pharmacies Grow Rapidly:
Hospital Pharmacies segment dominated the market among the distributors of rapid diagnostic kits due to the excellent procurement process and delivery of products to hospitals and diagnostics centers directly. Regular procurement and integration with healthcare services make them the preferred choice in the market.
Online Pharmacies are the fastest-growing segment, due to the increased digitization and popularity of online platforms and consumer inclination toward home diagnosis options. Better convenience, timely delivery, and increased faith in online healthcare platforms are contributing immensely.
Rapid Diagnostic Tests Market Regional Analysis:
North America Rapid Diagnostic Tests Market Insights:
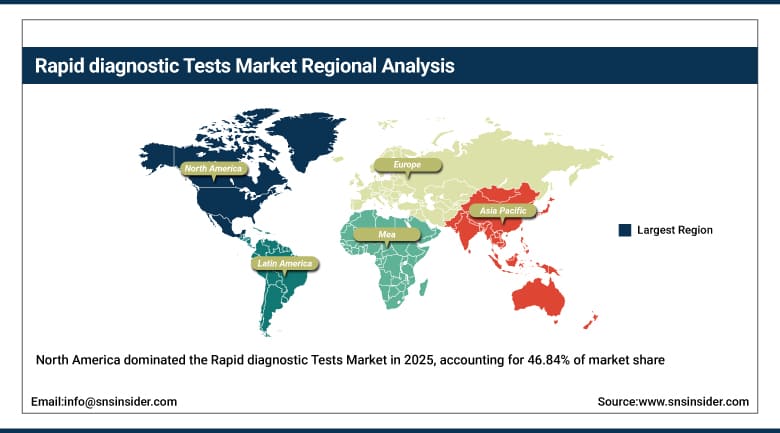
The North America Rapid Diagnostic Tests Market is dominated, holding a 46.84% share in 2025, fueled by the presence of sophisticated healthcare facilities and widespread adoption of rapid test technology in the United States and Canada. The high need for fast testing procedures in cases of infectious diseases diagnosis, emergencies, and routine screenings ensures that the demand for rapid tests will be consistent. In addition, frequent development of molecular rapid tests, digital diagnosis tests, and home testing options help boost market growth. The well-developed reimbursement system, dominant presence of key players, and high level of market awareness continue to contribute to North America’s dominance in this highly developed market, which continues to advance thanks to ongoing innovations.
Get Customized Report as Per Your Business Requirement - Enquiry Now
U.S. Rapid Diagnostic Tests Market Insights:
High testing rates, advanced diagnostic facilities, and widespread adoption of rapid test technology in hospitals, laboratories, and homes drive the U.S. Rapid Diagnostic Tests Market. Growing need for early diagnosis, use of self-testing kits, and incorporation of digital health care platforms help expand the market. The presence of leading companies and innovations in product development ensures the leadership of the region in the market.
Asia-Pacific Rapid Diagnostic Tests Market Insights:
The Asia-Pacific Rapid Diagnostic Tests Market is the fastest-growing region, projected to expand at a CAGR of 9.74% during 2026–2035. It is backed by the increasing prevalence of infectious diseases and improved healthcare services in China, India, Japan, and Southeast Asia. The increasing use of rapid diagnostic tests in both urban and rural health clinics, and the government's implementation of screening campaigns, will boost the rate of adoption. The rise in investments in diagnostic technology, increased recognition of the importance of early disease detection, and the availability of low-cost testing solutions will fuel substantial market growth within the region, aided by improved healthcare policies and increased private sector involvement.
China Rapid Diagnostic Tests Market Insights:
The China Rapid Diagnostic Tests Market is driven by a high burden of infectious diseases, expanding public health programs, and rapid development of healthcare infrastructure. Strong adoption of point-of-care testing, increasing use of molecular diagnostics, and large-scale screening initiatives across urban and rural regions are accelerating test utilization. Government support, domestic manufacturing capabilities, and rising investments in diagnostic innovation are positioning China as a major contributor to Asia-Pacific market growth.
Europe RDT Market Insights:
The Europe Rapid Diagnostic Tests market is characterized by efficient healthcare frameworks, rising significance on early disease diagnosis, and investments made in diagnostic technology. The countries such as Germany, France, and the UK will influence the regional demand due to the use of rapid tests in hospitals and laboratories. The expansion in the distribution of point-of-care testing, strict regulations, and reimbursement policies are contributing toward market growth. Developments in molecular diagnostics, technological advancements, and medical practitioners' awareness about RDTs enhance the Europe RDT market share.
Germany RDT Market Insights:
The Germany Rapid Diagnostic Tests market is an important segment within the European RDT market due to the presence of advanced diagnostic technologies and high priority given to disease detection. Growth will be propelled by rising acceptance of rapid tests, expansion of laboratories, and attention to quality and regulatory measures. Innovations in healthcare and diagnostic technologies bolster Germany's market position.
Latin America Rapid Diagnostic Tests Market Insights:
The Latin America Rapid Diagnostic Tests Market is growing due to rising incidence of infectious diseases and improving access to healthcare services. Adoption is supported by increasing use of rapid testing kits across Brazil, Mexico, and Argentina, along with expanding diagnostic programs and government initiatives. Investments in healthcare infrastructure and awareness campaigns are driving regional market growth, alongside rising demand for affordable diagnostics and expansion of private healthcare providers.
Middle East and Africa Rapid Diagnostic Tests Market Insights:
The Middle East & Africa Rapid Diagnostic Tests Market is expanding due to increasing focus on disease surveillance and improving healthcare infrastructure. Growing adoption of rapid testing solutions in hospitals and decentralized settings is driving demand, with Saudi Arabia, the UAE, and South Africa emerging as key regional markets. Government initiatives and international health programs are further supporting market expansion, along with increasing partnerships, funding support, and adoption of mobile healthcare services.
Rapid Diagnostic Tests Market Competitive Landscape:
Abbott Laboratories is one of the most successful healthcare firms headquartered in the United States and operates in the area of Rapid Diagnostic Tests with a variety of point-of-care and rapid test products including those for the detection of infections, heart ailments, and molecular diseases. Abbott Laboratories is known for its successful platforms that include BinaxNOW and ID NOW. The company's emphasis on rapid testing development, decentralized diagnostics, and digitalization of its processes has contributed to its dominant position in the industry.
-
In January 2025, Abbott expanded its rapid diagnostics portfolio with new point-of-care respiratory panels, enhancing multi-disease detection capabilities and strengthening its presence in decentralized testing environments.
F. Hoffmann-La Roche Ltd. is a prominent Swiss diagnostics leader with extensive expertise in molecular diagnostics and rapid testing solutions, particularly through its cobas and lateral flow platforms. The company focuses on high-precision diagnostic technologies, integrating rapid testing with advanced laboratory systems to support accurate disease detection and monitoring. Roche’s strong emphasis on innovation, personalized diagnostics, and healthcare partnerships supports its leadership in infectious disease testing and critical care diagnostics. Its robust R&D pipeline and integrated diagnostics approach continue to drive growth in the RDT market.
-
In March 2025, Roche introduced an advanced rapid molecular testing solution for infectious diseases, improving turnaround time and diagnostic accuracy, and expanding its point-of-care testing capabilities.
Siemens Healthineers is an international company in the medical technology industry that continues to establish itself in the Rapid Diagnostic Tests (RDT) market through advanced Point of Care (POC) and Near Patient Testing (NPT). The firm develops rapid immunoassay systems and diagnostics solutions intended to provide high efficiency, accuracy, and quick turnaround time. In addition, the organization invests heavily in research and development, strategic partnerships, and decentralized testing systems to strengthen its position in the RDT market environment.
-
In February 2025, Siemens Healthineers enhanced its point-of-care diagnostics portfolio with upgraded rapid testing platforms, improving workflow efficiency and expanding access to fast diagnostic solutions across healthcare settings.
Rapid diagnostic tests Market Key Players:
Some of the Rapid diagnostic tests Market Companies are:
-
Abbott Laboratories
-
F. Hoffmann-La Roche Ltd.
-
Siemens Healthineers AG
-
Danaher Corporation
-
Becton, Dickinson and Company (BD)
-
Thermo Fisher Scientific Inc.
-
bioMérieux SA
-
QuidelOrtho Corporation
-
SD Biosensor Inc.
-
Access Bio, Inc.
-
OraSure Technologies, Inc.
-
Chembio Diagnostics, Inc.
-
Trinity Biotech plc
-
Sekisui Diagnostics
-
Meridian Bioscience, Inc.
-
ACON Laboratories, Inc.
-
CTK Biotech, Inc.
-
Wondfo Biotech Co., Ltd.
-
Guangzhou Healgen Scientific LLC
-
LumiraDx Limited
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 32.88 Billion |
| Market Size by 2035 | USD 67.83 Billion |
| CAGR | CAGR of 7.62% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Product Type (Infectious Disease RDTs, Cardiac Marker RDTs, Pregnancy & Fertility RDTs, Glucose Monitoring RDTs, Others) • By Technology (Lateral Flow Immunoassays (LFIA), Molecular Rapid Tests, Agglutination Assays, Others) • By Application (Infectious Disease Testing, Cardiology Testing, Pregnancy & Fertility Testing, Blood Glucose Monitoring, Others) • By End User (Hospitals & Clinics, Diagnostic Laboratories, Home Care Settings, Others) • By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Direct Tenders (Government & NGOs), Others) |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
| Company Profiles | Abbott Laboratories, F. Hoffmann-La Roche Ltd., Siemens Healthineers AG, Danaher Corporation, Becton, Dickinson and Company (BD), Thermo Fisher Scientific Inc., bioMérieux SA, QuidelOrtho Corporation, SD Biosensor Inc., Access Bio, Inc., OraSure Technologies, Inc., Chembio Diagnostics, Inc., Trinity Biotech plc, Sekisui Diagnostics, Meridian Bioscience, Inc., ACON Laboratories, Inc., CTK Biotech, Inc., Wondfo Biotech Co., Ltd., Guangzhou Healgen Scientific LLC, LumiraDx Limited |

