Zika Virus Testing Market Report Scope & Overview:
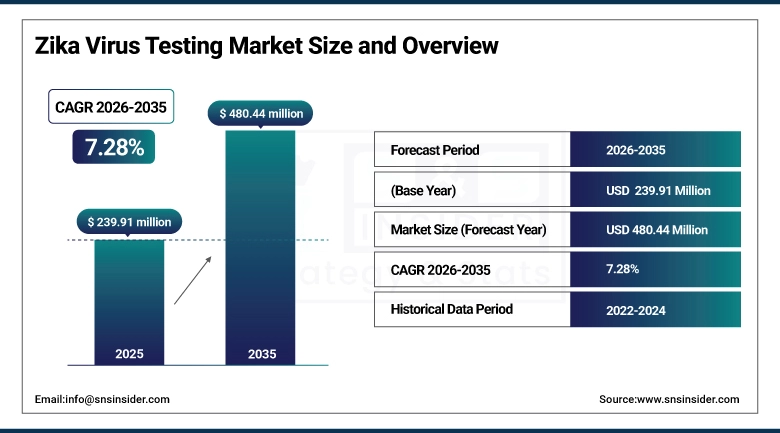
The Zika Virus Testing Market size is valued at USD 239.91 Million in 2025 and is projected to reach USD 480.44 Million by 2035, growing at a CAGR of 7.28% during the forecast period 2026–2035.
The analysis report on Zika Virus Testing Market offers in-depth analysis with respect to market dynamics, drug developments, and therapeutic uses. The increase in organ transplants, prevalence of autoimmune and inflammatory diseases, use of biologics, and improvement in healthcare infrastructure are some factors contributing to the high growth rate in the Zika Virus Testing Market from 2026 to 2035.
The use of Zika Virus Testing kit exceeded 1.2 billion prescriptions in 2025, mainly due to the increase in organ transplants and autoimmune diseases.
Market Size and Forecast:
-
Market Size in 2025: USD 239.91 Million
-
Market Size by 2035: USD 480.44 Million
-
CAGR: 7.28% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get more information on Zika Virus Testing Market - Request Free Sample Report
Zika Virus Testing Market Trends:
-
Testing volume increases during epidemic periods in Latin America and Asia-Pacific, followed by stabilization during inter-epidemic periods.
-
Increasing market acceptance of multiplex PCR tests that simultaneously detect Zika, dengue, and chikungunya viruses.
-
Portable rapid test kits are gaining popularity for rural settings and epidemic-prone regions.
-
EUA clearance facilitates faster market introduction during epidemic periods.
-
Increasing need for field-deployable PCR platforms for mobile health initiatives.
-
Increasing Zika testing as part of prenatal care packages in Zika-endemic regions.
-
Automated reporting systems directly interface diagnostic results to national databases or WHO/PAHO surveillance systems.
-
Miniaturization trends for paediatric and neonatal testing.
U.S. Zika Virus Testing Market Insights:
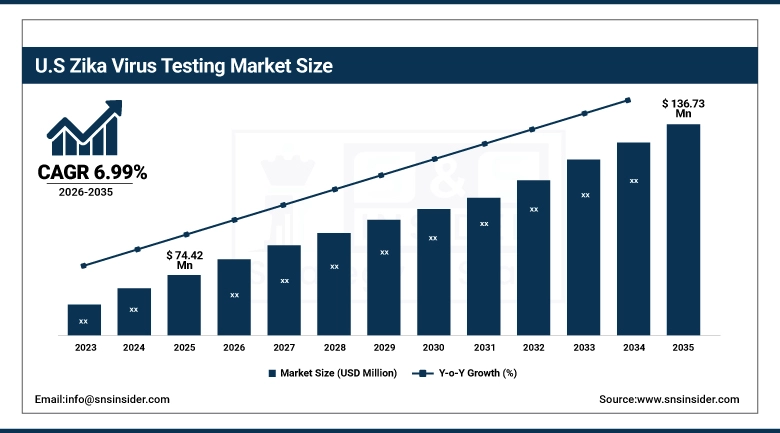
The US Zika Virus Testing Market is estimated to rise from USD 74.42 million in 2025 to USD 136.73 million in 2035 at a CAGR of 6.99%. The market is influenced by increased preparedness for Zika virus outbreaks, increased testing needs for prenatal and neonatal diagnosis, strong acceptance of advanced molecular testing technologies and multiplex testing solutions, and investments in novel rapid, portable, and deployable testing solutions in hospital settings, laboratories, and government agencies.
Zika Virus Testing Market Growth Drivers:
-
Rising outbreak preparedness and increasing demand for prenatal and neonatal screening are key drivers of Zika Virus Testing Market growth.
Healthcare organisations like Hospitals, diagnostic labs are increasingly using advanced molecular diagnostics, multiplex technologies, and rapid point of care kits to diagnose Zika Virus, prevent congenital problems, and control the outbreak. Advances in portable PCR technologies, rapid kits, and automated surveillance reporting are further propelling the use of these technologies to diagnose Zika Virus, increasing the accuracy of the tests, and fueling the growth of this market.
In 2025, over 58% of hospitals, diagnostic labs, and public health organizations used advanced Zika Virus Testing.
Zika Virus Testing Market Restraints:
-
High dependency on outbreak frequency and limited awareness in non-endemic regions are key restraints for Zika Virus Testing Market growth.
Public health organizations may have varying needs, with testing needs peaking during epidemics and dropping off significantly during inter-epidemic periods. Issues with regulatory approval, inconsistent funding for mosquito-borne diseases, as well as infrastructure challenges in rural or resource-constrained environments, are some of the challenges to market expansion. Difficulty in differentiating Zika from other arboviruses like dengue or chikungunya, as well as variability in test sensitivity and specificity, are some of the issues that affect test results.
In 2025, fewer than 42% of hospitals and diagnostic labs in non-endemic regions consistently offered Zika Virus Testing.
Zika Virus Testing Market Opportunities:
-
Expanding prenatal and neonatal screening programs and rising investments in multiplex molecular diagnostics present significant opportunities for Zika Virus Testing Market growth.
Healthcare infrastructure has begun to incorporate Zika virus testing as a standard procedure in their maternal healthcare packages, which has significantly enhanced the chances of early detection and prevention of birth-related complications. The availability of advanced portable PCR technology, quick point-of-care kits, and deployable diagnostic systems has also created new opportunities, especially in rural and endemic regions. The integration of reporting systems has also enhanced surveillance capabilities.
In 2025, more than 62% of public health organizations and hospitals in endemic regions recognized Zika Virus Testing as a strategic priority in outbreak preparedness and protection of maternal health.
Zika Virus Testing Market Segmentation Analysis:
-
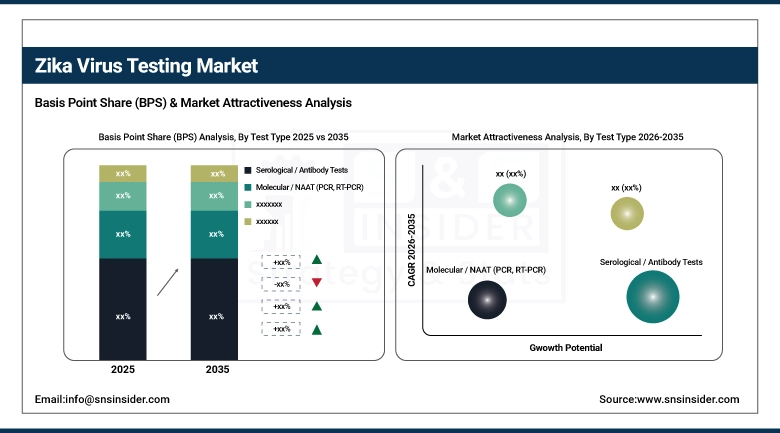
By Test Type, Serological / Antibody Tests held the largest market share of 61.46% in 2025, while Molecular / NAAT (PCR, RT-PCR) are expected to grow at the fastest CAGR of 7.41% during 2026–2035.
-
By Technology, Nucleic Acid Amplification (PCR, RT-PCR, qPCR) dominated with 46.55% market share in 2025, as they are projected to record the fastest CAGR of 6.79% through 2026–2035.
-
By Application, Clinical Diagnosis accounted for the highest market share of 49.12% in 2025, while Public Health Surveillance & Outbreak Control are expected to grow at the fastest CAGR of 6.71% during the forecast period.
-
By End User, Hospitals & Clinics held the largest share of 47.72% in 2025, while Diagnostic Units / Pathology Labs are expected to grow at the fastest CAGR of 6.82% during the forecast period.
By Test Type, Serological / Antibody Tests Dominate While Molecular / NAAT (PCR, RT-PCR) Grow Rapidly:
The Serological / Antibody Tests segment held the highest share in the market, primarily due to the high volume of tests conducted in large-scale screening programs, cost-effectiveness, and existing clinical acceptance in hospitals, labs, and health organizations. The testing volume exceeded 120 million tests in the upcoming years, indicating a strong preference among healthcare providers for screening tests.
Molecular / NAAT (PCR, RT-PCR) is the fastest-growing segment, primarily due to the growing need for accurate diagnostic tests, prenatal and neonatal screening, and the development of multiplex tests to simultaneously detect Zika, dengue, and chikungunya. The testing volume grew significantly, with over 65 million tests conducted, particularly in regions prone to outbreaks, indicating the trend towards the use of advanced molecular tests to accurately detect the virus.
By Technology, Nucleic Acid Amplification (PCR, RT-PCR, qPCR) Dominates While Also Growing Rapidly:
Segment-wise, the Nucleic Acid Amplification segment held the largest share of the market, owing to its accuracy, reliability, and acceptance by hospitals, diagnostic centers, and public health authorities. In 2025, the number of tests performed crossed the 95 million mark, which indicates the acceptance of this technology by healthcare providers, as they prefer high-end molecular diagnostic tools that can detect low levels of the virus with accuracy.
Moreover, the Nucleic Acid Amplification technology holds the position of the fastest-growing segment of the Zika Virus Testing Market, owing to the increasing need for multiplexing tests for the simultaneous detection of Zika, dengue, and chikungunya, as well as advancements in portable PCR systems, which have seen a surge in adoption, especially for prenatal, perinatal, and outbreak-prone areas, thus proving the dual functionality of PCR as the dominant as well as the fastest-growing technology of the Zika Virus Testing Market.
By Application, Clinical Diagnosis Dominates While Public Health Surveillance & Outbreak Control Grow Rapidly:
The Clinical Diagnosis segment accounted for the highest share of the market due to the use of tests in hospitals, clinics, and diagnostic labs for symptomatic patients as well as prenatal screening. The tests have surpassed the 140 million assay mark in 2025, indicating the strong acceptance of tests by healthcare providers to diagnose patients at an early stage of infection to prevent congenital problems.
The Public Health Surveillance & Outbreak Control segment of the market is growing at the fastest pace due to increasing government initiatives, WHO/PAHO-endorsed programs, and the need to quickly respond to epidemics. The tests have witnessed significant adoption, with over 85 million tests conducted in 2025 in outbreak-prone regions.
By End User, Hospitals & Clinics Dominate While Diagnostic Units / Pathology Labs Grow Rapidly:
The Hospitals & Clinics segment accounted for the highest share of the market due to their importance in the management of patients presenting with symptoms, prenatal screening, and outbreak response. The number of tests surpassed 300 million tests by the end of 2035, indicating the strong preference of healthcare providers for hospital-based tests that provide immediate care to patients.
Diagnostic Units / Pathology Labs are expected to be the fastest-growing segment of the market due to the increasing need for specialized molecular tests, the use of multiplex tests, and the increasing capacity to support large-scale screening during epidemics. This segment saw a significant increase in adoption, with over 90 million tests carried out in 2025, especially in regions prone to epidemics.
Zika Virus Testing Market Regional Analysis:
North America Zika Virus Testing Market Insights:
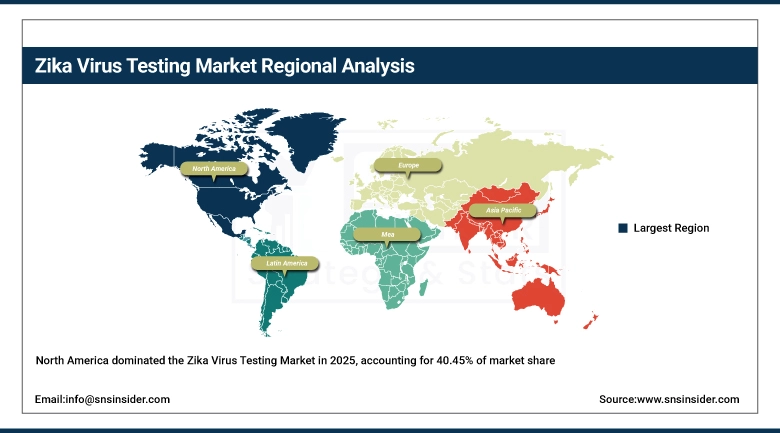
Under the North America Zika Virus Testing Market segment, there is a dominant market share of 40.45%, which is substantial in 2025, owing to the strong health infrastructure of the United States and Canadian countries. This has been made possible through a number of travel-related screenings, which are conducted, along with advancements made through molecular diagnostic tools, including Zika virus testing as a part of prenatal care.
Get Customized Report as per Your Business Requirement - Enquiry Now
U.S. Zika Virus Testing Market Insights:
The driving factors in the U.S. Zika Virus Testing Market include the strong testing infrastructure, high testing demand for travelers, and the strong testing demand for molecular tests in hospitals and clinics. The emphasis on prenatal and neonatal testing, the growing diagnostic labs, and government initiatives have helped the U.S. to hold the highest share in the North American region. The investments in multiplex assays and portable PCR have helped to combat the Zika virus.
Asia-Pacific Zika Virus Testing Market Insights:
Asia-Pacific has the fastest growth rate with a CAGR of 8.84%, fueled by a high mosquito density, increased outbreak risks, and growing government-initiated screening programs. Countries like India, Southeast Asia, and certain countries in Oceania drive this growth. Hospitals are increasingly using multiplex PCR tests, whereas portable rapid kits are gaining popularity, especially in rural areas with high outbreak risks. High public health investments, along with strong collaborations with international organizations, have helped APAC achieve its high growth rate.
China Zika Virus Testing Market Insights:
The China Zika Virus Testing market is growing at a rapid pace, driven by increased initiatives taken by the government to enhance infectious disease surveillance and maternal healthcare. Hospitals and labs are increasingly using molecular tests and multiplex PCR technology to test for the presence of Zika, dengue, and chikungunya. An increase in investment in portable PCR technology and quick point of care kits is also a driving force in the China market. The strong public health infrastructure, along with partnerships with international organizations, makes China a promising market in the Asia-Pacific region.
Europe Zika Virus Testing Market Insights:
The market for Europe is influenced by travel-related screening and preventive diagnostics, especially in endemic areas with high inbound travel. Hospitals and labs use molecular assays for their accuracy, whereas government agencies focus on integration with surveillance. Favorable regulatory environments and investments in sophisticated laboratory technologies contribute to the growth of the market.
Germany Zika Virus Testing Market Insights:
Germany Zika Virus Testing Market: The major factor influencing the Zika Virus Testing Market in Germany is related to travel, as a large number of tourists visit the country, especially from Zika-affected areas. In hospitals, there is a focus on molecular diagnostic tests, as they are more accurate, while health authorities focus on arbovirus surveillance, which includes Zika virus testing. The country has a conducive environment for conducting Zika virus tests, with well-equipped laboratories and a high adoption of multiplex technology.
Latin America Zika Virus Testing Market Insights:
Latin America is driving the global market due to the high occurrence rate and high disease burden. Brazil, Mexico, and Colombia are at the forefront in embracing the technology, and it has been widely integrated in hospitals and public health authorities. Government programs, WHO/PAHO partnerships, and the use of rapid kits have been instrumental in driving the market. Advances in multiplex technology and portable PCR systems are vital in containing the outbreak.
Middle East and Africa Zika Virus Testing Market Insights:
The Middle East and Africa are smaller markets that are growing steadily, with use concentrated in outbreak-prone areas and travel hubs. Public health organizations are focusing on surveillance and rapid kit deployment, while hospitals are using molecular tests for prenatal screening. The region still faces infrastructural and funding constraints, but aid programs and portable tests are helping.
Zika Virus Testing Market Competitive Landscape:
Roche Holding AG is a prominent Swiss multinational healthcare group with a strong footprint in the Zika Virus Testing industry. Its strong position in the market can be attributed to its cutting-edge molecular diagnostics technology, including cobas PCR systems. Additionally, its strong R&D efforts in developing multiplexed assays for the simultaneous detection of Zika virus along with dengue and chikungunya ensure high sensitivity and specificity. Its global manufacturing capabilities and lifecycle management strategies with automated reporting systems further add to its competitive advantage.
-
In September 2025, Roche announced the launch of a next‑generation multiplex PCR kit designed for simultaneous detection of arboviruses, enhancing outbreak preparedness and maternal health screening.
Abbott Laboratories is one of the largest healthcare companies in the US with a significant presence in the field of infectious disease testing. Their products in the category of ‘Zika Virus Testing’ include the ‘i-STAT’ and ‘m2000’ platforms. Both these platforms have gained significant traction in hospitals and labs across the country. The company’s leadership in the field is also a result of their innovative portable ‘point of care kits.’ They have also gained significant traction in the field.
-
In July 2025, Abbott introduced a portable Zika rapid test kit with enhanced reagent stability for tropical climates, aimed at expanding access in Latin America and Asia-Pacific.
Thermo Fisher Scientific Inc. is a US-based global leader in laboratory diagnostics and life sciences, possessing a strong portfolio in PCR reagents, ELISA kits, and multiplex assay systems for Zika Virus Testing. The company’s market position can be attributed to its strong relationships with public health organizations, reagent manufacturing capabilities, and its innovations in portable molecular technologies. Thermo Fisher’s focus on miniaturization and detection of low viral loads has further solidified its position as a market leader.
-
In November 2025, Thermo Fisher launched a mobile PCR platform optimized for rural and resource‑constrained settings, enabling faster outbreak containment and maternal health protection.
Zika Virus Testing Market Key Players:
Some of the Zika Virus Testing Market Companies are:
-
Roche
-
Abbott Laboratories
-
Thermo Fisher Scientific
-
Quest Diagnostics
-
ARUP Laboratories
-
Eurofins Scientific
-
DiaSorin
-
Chembio Diagnostics
-
Hologic
-
Luminex Corporation
-
Siemens Healthineers
-
Altona Diagnostics
-
Creative Diagnostics
-
InBios International
-
Biocan Diagnostics
-
QIAGEN
-
bioMérieux
-
Bio-Rad Laboratories
-
Access Bio
-
Becton Dickinson (BD)
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 239.91 Million |
| Market Size by 2035 | USD 480.44 Million |
| CAGR | CAGR of 7.28% From 2026 to 2033 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Test Type(Serological/Antibody Tests, Molecular / NAAT (PCR, RT-PCR), Rapid Diagnostic Kits (EUA-based), Others) • By Nucleic Acid Amplification (PCR, RT-PCR, qPCR), ELISA / Serological Assays, Point-of-Care Rapid Kits, Others) • By Application (Clinical Diagnosis, Public Health Surveillance & Outbreak Control, Travel & Preventive Screening, Research & Academic Studies, Others) • ByEnd User (Hospitals & Clinics, Diagnostic Units / Pathology Labs, Research Institutes, Others) |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
| Company Profiles | Roche, Abbott Laboratories, Thermo Fisher Scientific, Quest Diagnostics, ARUP Laboratories, Eurofins Scientific, DiaSorin, Chembio Diagnostics, Hologic, Luminex Corporation, Siemens Healthineers, Altona Diagnostics, Creative Diagnostics, InBios International, Biocan Diagnostics, QIAGEN, bioMérieux, Bio-Rad Laboratories, Access Bio, Becton Dickinson (BD). |
Frequently Asked Questions
Ans: Key drivers include outbreak preparedness, prenatal and neonatal screening programs, adoption of multiplex molecular assays, and innovations in portable point-of-care kits.
Ans: Latin America leads due to frequent outbreaks, while Asia-Pacific is the fastest-growing region. North America and Europe see steady demand from travel-related screening and preventive diagnostics.
Ans: Key challenges include outbreak dependency, low awareness in non-endemic areas, regulatory delays, rural infrastructure limits, and difficulty distinguishing Zika from similar arboviruses.
Ans: Prenatal and neonatal screening to prevent congenital Zika syndrome.
Ans: The primary test types include Serological/Antibody Tests for large-scale screening and Molecular/NAAT (PCR, RT-PCR, qPCR) assays for highly accurate detection of active infections.

