Cosmetovigilance Market Report Scope & Overview:
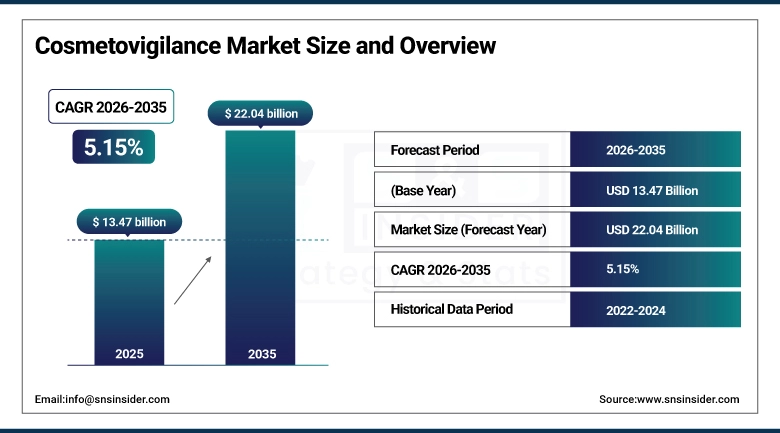
The Cosmetovigilance Market size is valued at USD 13.47 Billion in 2025 and is projected to reach USD 22.04 Billion by 2035, growing at a CAGR of 5.15% during the forecast period 2026–2035.
Cosmetovigilance Market Analysis Report presents an in-depth evaluation of market dynamics, service innovation, and regulatory application. The increasing need for skin care and personal care products, rising consumer awareness about product safety, the usage of AI-based monitoring devices, and the widening scope of regulatory frameworks have positively influenced the market's dynamic growth over 2026-2035.
The application of both pre-market and post-market safety services crossed USD 3.5 billion in 2025, wherein pre-market compliance holds the lion's share.
Market Size and Forecast:
-
Market Size in 2025: USD 13.47 Billion
-
Market Size by 2035: USD 22.04 Billion
-
CAGR: 5.15% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get more information on Cosmetovigilance Market - Request Free Sample Report
Cosmetovigilance Market Trends:
-
Increased application of artificial intelligence-based signal detection for detecting adverse effects via social media, online reviews, and consumer feedback.
-
Surging interest in skincare products category surveillance due to the increased regulatory concern about anti-aging and sensitive skin products.
-
Increased contribution of dermatologists' offices and hospitals in the process of reporting adverse effects of cosmetic products.
-
Increased application of electronic tools in the processes of making regulatory submissions, tracking adverse events, and compliance reporting.
-
Increased value of toxicological risk assessment and labeling validation for ensuring that the statements made on the labels are science-based.
-
Moving toward preventive approaches by incorporating corrective and preventive action (CAPA) systems in the process of recalls and reformulation.
U.S. Cosmetovigilance Market Insights:
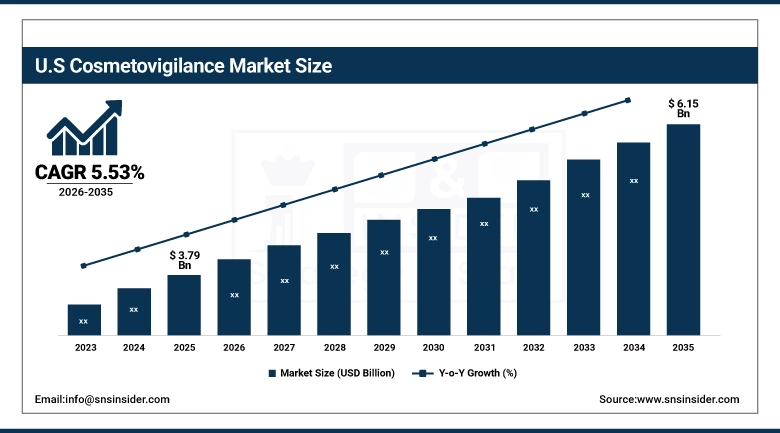
The US Cosmetovigilance Market is expected to increase from USD 3.79 billion in 2025 to USD 6.15 billion in 2035, growing at a CAGR of 5.53%. The market will be boosted by growing demands for skincare and personal care products, increased consumer consciousness regarding the safety of these products, high acceptance of AI-based surveillance systems, and advancements in pre marketing and post marketing compliance solutions.
Cosmetovigilance Market Growth Drivers:
-
Rising consumer awareness and demand for product safety are driving adoption of advanced cosmetovigilance services.
Growth is driven largely by issues related to adverse events and disclosure requirements. There has been an increasing trend toward the implementation of pre-market and post-market safety services by cosmetic manufacturers and regulatory bodies through the use of artificial intelligence-driven monitoring systems and software. Automation of reporting, real-time signaling, and harmonized regulations have also been instrumental in driving the market ahead.
In excess of 60% of cosmetic manufacturing companies and regulatory bodies have employed cosmetovigilance platforms in 2025.
Cosmetovigilance Market Restraints:
-
High implementation costs and limited budgets are restraining adoption of advanced cosmetovigilance platforms.
Small cosmetic companies and new entrants usually find it difficult to invest enough in pre marketing and post marketing safety processes. The requirement for technical knowledge, investments in artificial intelligence technology, and compliance with various regulatory requirements make operations expensive, discouraging smaller companies from investing.
By 2025, more than 40% of small and medium cosmetic producers cited cost challenges as the main factor deterring them from investing in cutting edge cosmetovigilance platforms.
Cosmetovigilance Market Opportunities:
-
Integration of AI and big data analytics is creating new opportunities for advanced cosmetovigilance.
There is great scope to incorporate automated systems that can detect any adverse events through various sources including social media, ecommerce reviews, and dermatological findings to mitigate risks. The hybrid approach to outsourcing is presenting fresh opportunities to optimize performance. Organizations are now embracing collaborative strategies where the core processes are managed internally, and other tasks, including toxicological assessment and artificial intelligence surveillance, are outsourced.
By 2025, more than 48% of top-notch cosmetic companies had incorporated AI enabled monitoring systems, demonstrating immense prospects for technological growth.
Cosmetovigilance Market Segmentation Analysis:
-
By Service Type, Pre-Marketing Services held the largest market share of 56.38% in 2025, while Post-Marketing Services are expected to grow at the fastest CAGR of 5.80% during 2026–2035.
-
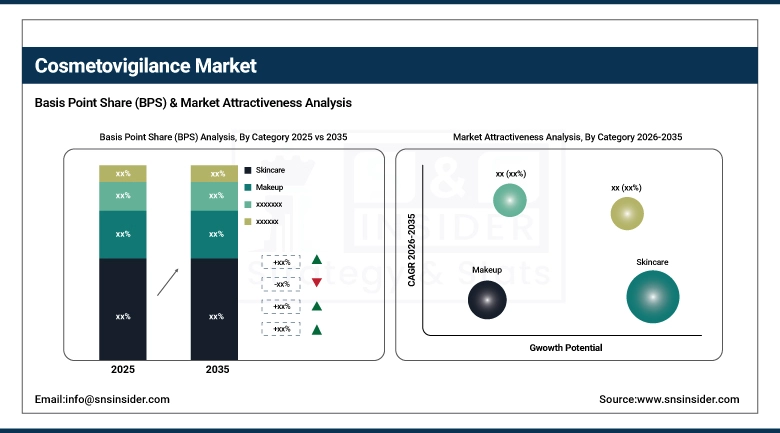
By Category, Skincare dominated with 38.44% market share in 2025, whereas they are also projected to record the fastest CAGR of 5.97% through 2026–2035.
-
By Service Provider, In-house accounted for the highest market share of 56.94% in 2025, while Contract Outsourcing are expected to grow at the fastest CAGR of 5.79% during the forecast period.
-
By End User, Cosmetic Manufacturers held the largest share of 57.51% in 2025, while Dermatology Clinics & Hospitals are expected to grow at the fastest CAGR of 7.42% during the forecast period.
By Category, Skincare Dominates While Also Growing Fastest:
The skincare category had the highest market share as it plays a crucial role in consumer safety surveillance especially in antiaging, sensitive skin and natural products. Both cosmetic companies and regulatory bodies give priority to skincare products as they have a higher rate of usage and more possibilities of developing allergies.
The skincare industry has witnessed an unprecedented number of innovations including probiotics, plant-based active ingredients and artificial intelligence for adverse event surveillance. This trend has played a major part in the growing popularity of skincare and makes it the biggest and most vibrant category in the cosmetics market.
By Service Type, Pre‑Marketing Services Dominate While Post‑Marketing Services Grow Rapidly:
The Pre Marketing Services segment led the market due to its significant contribution toward making sure that the cosmetic products adhere to all the safety and compliance measures prior to their release into the market. The manufacturers and the regulatory bodies have a lot of faith in the pre marketing processes such as toxicological risk assessment, label verification, and submission of the safety dossier.
Post Marketing Services is the most rapidly growing segment due to the increasingly stringent international guidelines, increased consumer demands, and the implementation of the Artificial Intelligence enabled surveillance systems.
By Service Provider, In‑House Models Dominate While Contract Outsourcing Expands Rapidly:
The In House service provider model was dominant due to big cosmetic manufacturers are keen on maintaining control over their operations in terms of safety, regulatory compliance, and other issues. These aspects are best handled using confidentiality and more streamlined processes, and hence, this became the most popular model.
On the other hand, Contract Outsourcing is the fastest-growing category. This is due to the increasing needs of specialized services at lower costs and scalability. Cosmetics companies are increasingly working together with outside service providers to perform toxicity assessments, utilize AI surveillance technology, and attain international regulatory compliance among others.
By End User, Hospitals Dominate While Specialty Clinics Grow Rapidly:
The hospitals market segment accounted for the highest share in 2025, serving as the leading place where organ transplantations and acute autoimmune reactions are managed. It has administered more than 580 million prescriptions in the year 2025 due to its high patient volume and quality pharmaceutical services. Growth in infrastructure and availability of specialist healthcare professionals has driven their adoption.
Among all other segments, the specialty clinics account for the highest growth rate, owing to rising preference for out-patient care, home-based therapies, and treatment of chronic autoimmune disorders. Specialty clinics have administered more than 220 million prescriptions in the year 2025.
Cosmetovigilance Market Regional Analysis:
Europe Cosmetovigilance Market Insights:
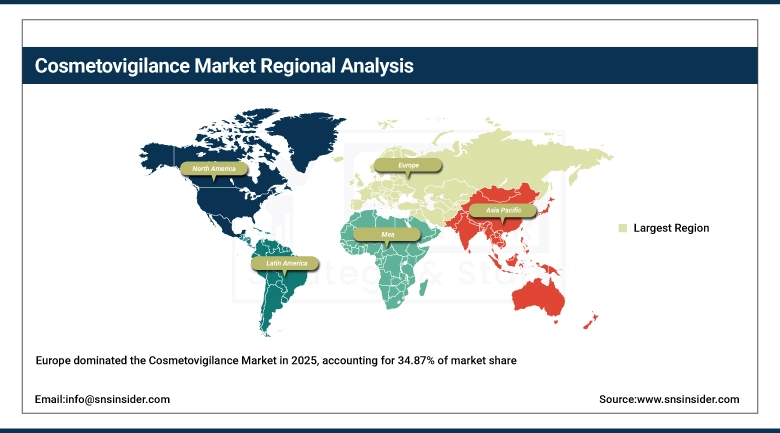
Europe has the largest share in the cosmetovigilance market with 34.87%, driven by EU laws, awareness among consumers about product safety, and systems for the monitoring of adverse effects. Europe has maintained its dominance due to the high level of acceptance among cosmetic companies of pre marketing and post marketing compliance systems. Innovation in skin care products and natural-based cosmetic formulations is contributing to the steady rise in the cosmetovigilance market.
Get Customized Report as per Your Business Requirement - Enquiry Now
Germany Cosmetovigilance Market Insights:
Germany takes the lead among other European countries owing to its superior health care facilities, expertise in dermatology, and stringent safety standards for products. The cosmetic companies operating in Germany place emphasis on conducting thorough assessments before bringing a new product to market and effective postmarketing surveillance, which ensures high compliance rates.
Asia‑Pacific Cosmetovigilance Market Insights:
Asia Pacific Region is witnessing high growth potential, recording a CAGR of 6.91% due to escalating cosmetics usage, an increase in middle-class population sizes, and changing regulatory policies. Japan, South Korea, and India are developing their safety surveillance programs to cater to growing needs for skin care and personal care products. The rising consumer concern for product safety, along with quick uptake of artificial intelligence-enabled surveillance solutions, will facilitate the market expansion.
China Cosmetovigilance Market Insights:
There have been tremendous developments within the cosmetovigilance market in China due to high demands for skin care and cosmetics. The NMPA has enforced stringent safety monitoring protocols that have made cosmetic companies adopt more compliant services. Cooperation between dermatology clinics/hospitals and cosmetics companies improves post-marketing surveillance, whereas increased adoption of AI-based monitoring solutions provides greater transparency.
North America Cosmetovigilance Market Insights:
The market in North America continues to be a developed one, due to the presence of good regulatory mechanisms, high-end healthcare facilities, and safety monitoring measures. The cosmetic producers and the regulatory authorities have their priorities well laid out regarding pre-marketing and post-marketing services. Increased demand for beauty products and the FDA ensure that there will always be an expanding market in North America. Cooperation between hospitals, dermatology clinics, and safety groups ensures effective adverse event monitoring in North America.
U.S. Cosmetovigilance Market Insights:
The U.S. cosmetic industry relies on good regulatory oversight, a very high consumer demand for safety in cosmetics, and widespread use of AI-based monitoring systems. The cosmetic companies are at the forefront when it comes to implementing rigorous compliance strategies prior to the launch of their products, and the post launch monitoring system is also expanding due to transparency obligations and efforts made by FDA. In addition, dermatology clinics and hospitals are crucial in reporting adverse events.
Latin America Cosmetovigilance Market Insights:
The cosmetovigilance market in Latin America continues to rise consistently due to increasing use of cosmetics by consumers in developing countries such as Brazil and Mexico and due to changing regulatory environments in these nations. With increased concern about product safety and demand for more natural products, cosmetic companies will find it increasingly necessary to adhere to stricter regulatory requirements. Coordinating with dermatological clinics and regulatory agencies helps with adverse event monitoring.
Middle East & Africa Cosmetovigilance Market Insights:
The Middle East & Africa region is still in the developmental phase, and its growth will be fueled by increased use of cosmetics, urbanization trends, and heightened consumer concern about the safety of products. Countries such as UAE and South Africa are implementing regulations that favor the use of pre-marketing and post-marketing services. Leading international cosmetics companies are penetrating the markets with advanced regulatory practices.
Cosmetovigilance Market Competitive Landscape:
Pharmalex
Pharmalex is a renowned service provider in areas such as regulation, pharmacovigilance, and cosmetovigilance. It assists pharmaceuticals, biotechnology, medical devices, and cosmetic industries in every phase of the product life cycle. Regulatory, compliance, safety, and risk management are key aspects that Pharmalex focuses on. The nature of the company and integration with Cencora makes the service provision even more efficient.
-
In March 2025, Pharmalex launched advanced automation tools for local PV monitoring and KPI tracking, improving efficiency and accuracy in cosmetovigilance reporting.
Freyr
Freyr is among the world's leading providers of regulatory solutions and services. The firm operates across more than 120 countries and employs over 2,500 regulatory experts. It focuses on providing regulatory intelligence, submission, labeling, artwork, and compliance solutions to both life science and cosmetic sectors. Its core offering, the AI-first solution Freya Fusion, combines regulatory information management and compliance monitoring. Freyr aims to ease compliance challenges and facilitate safe product launches in various regions around the world.
-
In August 2025, Freyr expanded its AI-driven Freya Fusion platform to include enhanced cosmetovigilance modules, enabling real-time adverse event signal detection and reporting across multiple geographies.
Arriello
Arriello is a regulatory affairs, pharmacovigilance, and compliance service provider based in Dublin. The company was founded in the year 2008 and caters to pharmaceuticals, biotechs, and cosmetics industries. Risk management, auditing, automation, and clinical safety management form some of the main services offered by the firm during the various stages of a product life cycle. The firm specializes in providing services relating to compliance with EU and US GxP regulations.
-
In April 2025, Arriello expanded its European operations with new automation-driven compliance services, strengthening its position in cosmetovigilance and regulatory risk management.
Cosmetovigilance Market Key Players:
Some of the Cosmetovigilance Market Companies are:
-
Pharmalex
-
Freyr
-
Arriello
-
Oviya MedSafe
-
Cliantha Research
-
UBC
-
PPD
-
BioPharma Services
-
MakroCare
-
AxPharma
-
Symogen
-
CRF Health
-
Clinevo
-
QPS
-
AssureSafety
-
Veeda Clinical Research
-
Accenture
-
PRA Health Sciences
-
Parexel
-
ICON Plc
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 13.47 Billion |
| Market Size by 2035 | USD 22.04 Billion |
| CAGR | CAGR of 5.15% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Service Type (Pre-Marketing Services, Post-Marketing Services, Others), • By Category (Skincare, Makeup, Haircare, Perfume & Deodorants, Others), • By Service Provider (In-house, Contract Outsourcing, Hybrid/Collaborative Models, Others), • By End User (Cosmetic Manufacturers, Regulatory Authorities, Dermatology Clinics & Hospitals, Consumer Safety Organizations, Others) |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
| Company Profiles | Pharmalex, Freyr, Arriello, Oviya MedSafe, Cliantha Research, UBC, PPD, BioPharma Services, MakroCare, AxPharma, Symogen, CRF Health, Clinevo, QPS, AssureSafety, Veeda Clinical Research, Accenture, PRA Health Sciences, Parexel, ICON Plc, Labcorp, Bioclinica, TFS International. |
Frequently Asked Questions
Ans: Skincare leads with 38.44% share and 5.97% CAGR, fueled by demand for anti-aging, sensitive skin, and natural formulations.
Ans: Cosmetic manufacturers dominate with 57.51% market share, followed by regulatory authorities, dermatology clinics, and consumer safety organizations.
Ans: Pre-marketing services hold the largest share of 56.38%, as every cosmetic product must undergo safety validation before launch, though post-marketing services are growing faster.
Ans: Asia-Pacific is the fastest-growing region with a CAGR of 6.91%, driven by booming cosmetics demand in India, China, and South Korea, alongside tightening regulatory frameworks.
Ans: The Cosmetovigilance Market is projected to grow at a CAGR of 5.15% during 2026–2035.

