Biomarker Testing Market Report Scope & Overview:
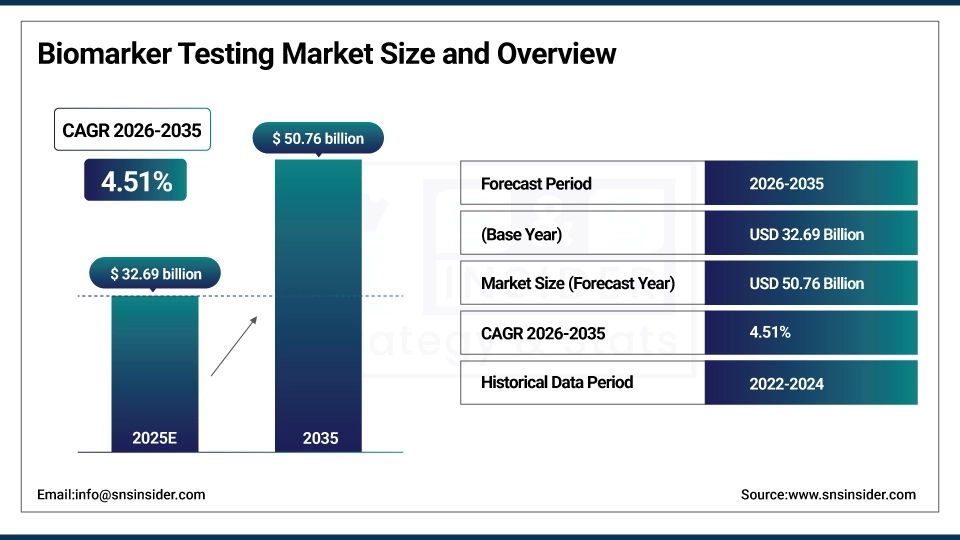
The Biomarker Testing Market size was valued at USD 32.69 Billion in 2025 and is projected to reach USD 50.76 Billion by 2035, growing at a CAGR of 4.51% during 2026–2035.
The global biomarker testing market is witnessing rapid growth due to the increasing demand for early disease detection, personalized treatment, and precision medicine. Biomarker testing enables healthcare providers to identify diseases at an early stage, monitor therapy response, and predict patient outcomes, improving overall healthcare efficiency. Advancements in molecular diagnostics, immunoassays, and high-throughput technologies are enhancing the accuracy, speed, and accessibility of testing. Additionally, rising investments in research and development, collaborations between pharmaceutical and diagnostic companies, and growing awareness among healthcare professionals and patients are fueling market expansion. The market is also benefiting from innovations such as non-invasive testing methods, liquid biopsies, and AI-driven analytics, which are transforming disease diagnosis and clinical decision-making.
Biomarker Testing Market Size and Forecast:
-
Market Size in 2025: USD 32.69 Billion
-
Market Size by 2035: USD 50.76 Billion
-
CAGR: 4.51% during 2026–2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get more information on Biomarker Testing Market - Request Free Sample Report
Biomarker Testing Market Trends:
-
Increasing adoption of personalized and precision medicine is driving demand for biomarker testing across oncology, cardiovascular, and neurological diseases.
-
Rapid advancements in molecular diagnostics and next-generation sequencing (NGS) are enabling faster, more accurate, and minimally invasive testing.
-
Growing focus on liquid biopsies and non-invasive biomarker detection is transforming early disease diagnosis and monitoring.
-
Expansion of hospital and diagnostic center networks worldwide is boosting accessibility and utilization of biomarker testing services.
-
Integration of AI and data analytics in biomarker research is accelerating biomarker discovery, clinical trial efficiency, and predictive disease modeling.
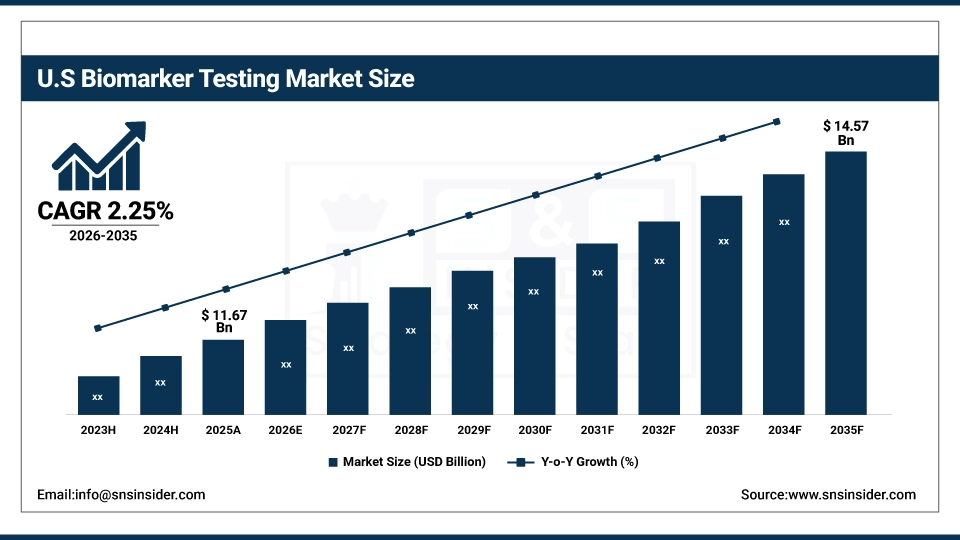
The U.S. Biomarker Testing Market is valued at USD 11.67 Billion in 2025 and is expected to reach USD 14.57 Billion in 2035, growing at a CAGR of 2.25% from 2026 to 2035. The U.S. Biomarker Testing Market growth is driven by rising chronic disease prevalence, adoption of precision medicine, advancements in molecular diagnostics, increased use of companion diagnostics, and growing clinical awareness and utilization.
Biomarker Testing Market Growth Drivers:
-
Rising prevalence of chronic diseases, personalized medicine adoption, and advancements in molecular diagnostics drive biomarker testing demand.
The major driver of the growth of the biomarker testing market is the increasing adoption of precision medicine and early disease detection tools across oncology, cardiovascular, and neurological disorders. Healthcare providers and research organizations are investing in genetic, protein, and molecular diagnostic testing to improve patient outcomes, therapy selection, and disease monitoring. The growing demand for companion diagnostics and targeted therapies is also propelling market expansion.
Biomarker Testing Market Restraints:
-
High costs, complex testing procedures, and regulatory hurdles may limit market adoption.
Challenges for the market include the high capital investment required for advanced biomarker testing equipment, reagents, and molecular platforms. Additional factors include the need for skilled professionals to conduct and interpret tests, stringent regulatory approvals, and concerns over data privacy and ethical use of genetic information, which may restrain adoption, especially in emerging markets and smaller healthcare facilities.
Biomarker Testing Market Opportunities:
-
Technological innovations, biomarker discovery, and rising demand for personalized therapies create growth avenues.
Opportunities exist in advanced molecular diagnostics, next-generation sequencing (NGS), immunoassays, and liquid biopsy technologies. Expanding research in oncology, cardiovascular, neurological, and rare diseases is driving the need for novel biomarkers. Collaborations between diagnostic companies, pharmaceutical firms, and research institutes, as well as increasing government funding for personalized medicine, are creating significant growth potential for market players.
Biomarker Testing Market Report Segment Analysis:
-
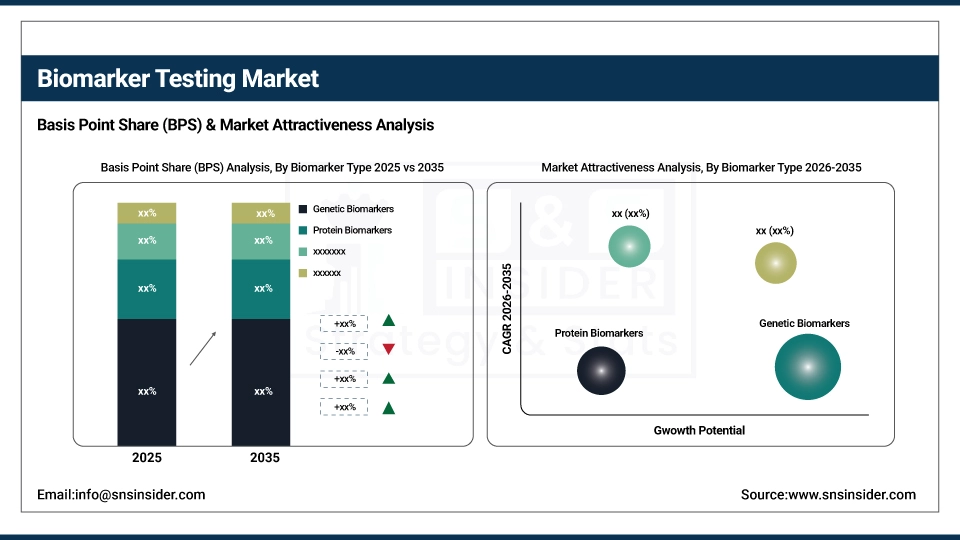
By Biomarker Type: In 2025, Genetic Biomarkers dominated with 40% share; Protein Biomarkers fastest growing segment during 2026-2035
-
By Technology: In 2025, Immunoassays dominated with 38% share; Molecular Diagnostics (PCR, NGS) fastest growing segment during 2026-2035
-
By End User: In 2025, Hospitals & Diagnostic Centers dominated with 50% share; Research & Academic Institutes fastest growing segment during 2026-2035
-
By Application: In 2025, Oncology dominated with 45% share; Cardiovascular & Neurological Disorders fastest growing segment during 2026-2035
By Biomarker Type: Genetic Biomarkers Dominates, Protein Biomarkers Fastest-Growing
Genetic biomarkers lead the biomarker type segment in the market, holding a share of approximately 40% in 2025, due to their crucial role in precision medicine, cancer diagnostics, and therapy selection. They enable accurate disease detection and targeted treatment, making them the largest contributor to the market.
Protein biomarkers are growing at a faster rate than other biomarker types, mainly due to their expanding applications in oncology, cardiovascular, and neurodegenerative disease detection.
By Technology: Immunoassays Dominates, Molecular Diagnostics Fastest-Growing
Immunoassays dominate the technology segment, accounting for around 38% of the market, driven by their reliability, cost-effectiveness, and widespread use in clinical laboratories. They are standard tools for routine biomarker detection, making them the largest contributor.
Molecular diagnostics (PCR, NGS) are growing at a faster pace due to their high sensitivity, precision, and rising adoption in personalized medicine and advanced research applications.
By End User: Hospitals & Diagnostic Centers Dominates, Research Institutes Fastest-Growing
Hospitals and diagnostic centers lead the end-user segment with a market share of roughly 50%, as they conduct the majority of biomarker tests for patient diagnosis and monitoring. Their established infrastructure makes them the primary market contributor.
Research and academic institutes are the fastest-growing end users, supported by increasing biomarker discovery, clinical trials, and demand for innovative diagnostic solutions.
By Application: Oncology Dominates, Cardiovascular & Neurological Disorders Fastest-Growing
Oncology dominates the application segment, holding approximately 45% of the market, due to the critical role of biomarker testing in early cancer detection, therapy selection, and treatment monitoring.
Cardiovascular and neurological disorders are the fastest-growing applications, driven by rising prevalence, early intervention needs, and growing focus on personalized treatment approaches.
Regional Analysis:
North America Biomarker Testing Market Insights:
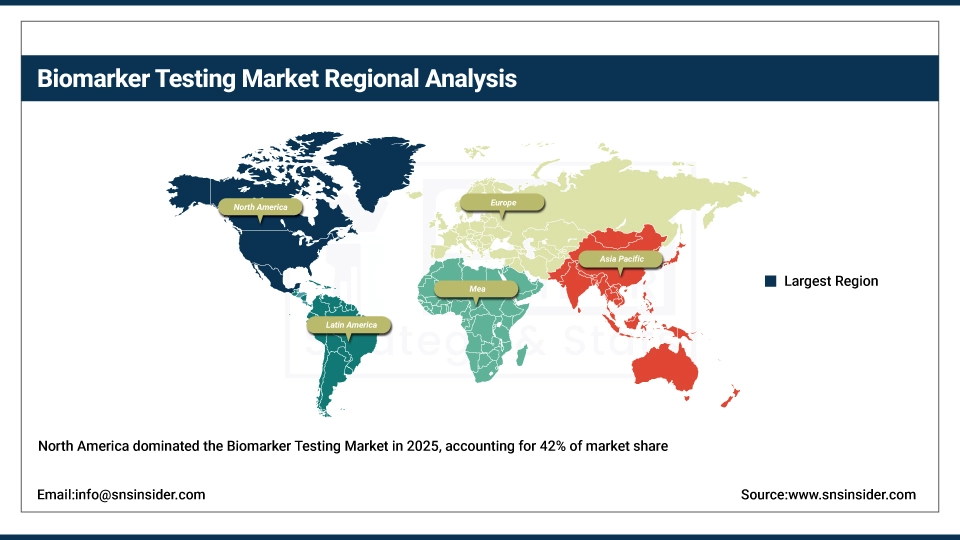
North America is the dominant region in the global biomarker testing market, accounting for approximately 42% market share in 2025. The region’s leadership is driven by advanced healthcare infrastructure, widespread adoption of molecular diagnostics, and significant investments in precision medicine and personalized therapies. High awareness among healthcare professionals and patients, along with strong research and development activities by key diagnostic and pharmaceutical companies, further bolster market growth in North America.
Get Customized Report as per Your Business Requirement - Enquiry Now
Europe Biomarker Testing Market Insights:
Europe is a significant market for biomarker testing, supported by established healthcare infrastructure, high adoption of advanced diagnostics, and growing focus on personalized medicine. Increasing prevalence of chronic diseases, oncology research, and government initiatives for early disease detection are driving steady demand. The market benefits from collaborations between diagnostic companies and research institutes, as well as ongoing technological advancements in molecular diagnostics, immunoassays, and non-invasive testing methods, fostering continuous growth across major European countries.
Asia-Pacific Biomarker Testing Market Insights:
Asia-Pacific is the fastest-growing region in the biomarker testing market, with a CAGR of 9.60% during 2026–2035. Growth is driven by rising healthcare investments, expanding diagnostic infrastructure, increasing awareness of precision medicine, and the adoption of advanced molecular and immunoassay-based testing. Emerging demand from hospitals, research institutes, and pharmaceutical companies, coupled with government initiatives supporting early disease detection and personalized therapies, is fueling rapid market expansion across countries like China, India, Japan, and Australia.
Latin America Biomarker Testing Market Insights:
Latin America is witnessing gradual growth in biomarker testing, driven by rising healthcare awareness, expanding diagnostic facilities, and increasing adoption of molecular and immunoassay-based testing. Government initiatives and partnerships are supporting early disease detection and personalized therapy programs.
Middle East & Africa (MEA) Biomarker Testing Market Insights:
The MEA biomarker testing market is emerging steadily, fueled by growing healthcare investments, rising awareness of early disease detection, and adoption of advanced diagnostic technologies. Expansion of hospital networks and government initiatives in personalized medicine are supporting market growth.
Biomarker Testing Market Competitive Landscape:
Roche Diagnostics is a Switzerland-based company founded in 1896, specializing in in-vitro diagnostics, molecular diagnostics, and personalized healthcare solutions. Roche develops biomarker testing solutions, companion diagnostics, and molecular assays for oncology, cardiovascular, and infectious disease applications worldwide.
Abbott Laboratories is a US-based company founded in 1888, specializing in diagnostics, medical devices, nutrition products, and branded generic pharmaceuticals. Abbott develops biomarker testing platforms, immunoassays, and point-of-care molecular diagnostics for oncology, cardiology, and infectious diseases worldwide.
-
In 2023, Abbott introduced the ARCHITECT i2000SR immunoassay system with expanded biomarker testing capabilities, improving diagnostic speed, accuracy, and accessibility across hospitals and clinical laboratories.
Biomarker Testing Market Key Players:
-
Roche Diagnostics
-
Abbott Laboratories
-
Thermo Fisher Scientific
-
QIAGEN N.V.
-
Danaher Corporation
-
Bio-Rad Laboratories, Inc.
-
Siemens Healthineers
-
Hologic, Inc.
-
Hoffmann-La Roche AG
-
Myriad Genetics, Inc.
-
Agilent Technologies, Inc.
-
PerkinElmer, Inc.
-
Becton, Dickinson and Company (BD)
-
GE Healthcare Life Sciences
-
Fluidigm Corporation
-
Illumina, Inc.
-
Bio-Techne Corporation
-
Oxford BioDynamics Ltd.
-
Guardant Health, Inc.
-
NanoString Technologies, Inc
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 32.69 Billion |
| Market Size by 2035 | USD 50.76 Billion |
| CAGR | CAGR of 4.51% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Biomarker Type: (Genetic Biomarkers, Protein Biomarkers, Cellular Biomarkers) • By Technology: (Immunoassays, Molecular Diagnostics (PCR, NGS), Flow Cytometry) • By End User: (Hospitals & Diagnostic Centers, Research & Academic Institutes, Pharmaceutical & Biotechnology Companies) • By Application: (Oncology, Cardiovascular Diseases, Neurological Disorders) |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
| Company Profiles | Roche Diagnostics, Abbott Laboratories, Thermo Fisher Scientific, QIAGEN N.V., Danaher Corporation, Bio-Rad Laboratories, Inc., Siemens Healthineers, Hologic, Inc., F. Hoffmann-La Roche AG, Myriad Genetics, Inc., Agilent Technologies, Inc., PerkinElmer, Inc., Becton, Dickinson and Company (BD), GE Healthcare Life Sciences, Fluidigm Corporation, Illumina, Inc., Bio-Techne Corporation, Oxford BioDynamics Ltd., Guardant Health, Inc., NanoString Technologies, Inc. |

