Red Biotechnology Market Report Scope & Overview:
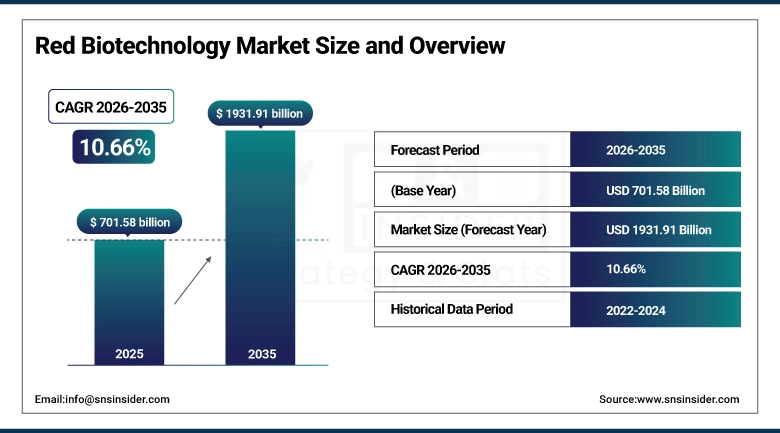
The Red Biotechnology Market size was valued at USD 701.58 billion in 2025 and is expected to reach USD 1,931.91 billion by 2035, growing at a CAGR of 10.66% over the forecast period of 2026-2035.
The global red biotechnology market trend is influenced by the increasing demand for biopharmaceutical products, including monoclonal antibodies, gene therapy products, and cell-based immunotherapy solutions, owing to the increasing incidence of chronic diseases, oncological disorders, and rare genetic disorders worldwide. Red biotechnology refers to the application of biotechnology for medical and pharmaceutical purposes. The growth trajectory for the red biotechnology market is revolutionized by the increasing discoveries related to CRISPR gene editing technologies, CAR-T cell therapies, mRNA platform technologies, and recombinant protein technologies. The increasing availability of bioprocessing infrastructure, drug discovery technologies through the application of artificial intelligence tools, and the availability of favorable regulations for biologics are supporting the growth trajectory for the red biotechnology market by the pharmaceutical and biotechnology industries.
For instance, in 2024, the FDA approved a record 12 biologics license applications (BLAs) in a single quarter, reflecting the surging clinical pipeline of biologic therapies and underscoring the accelerating commercialization pace within the red biotechnology sector globally.
Red Biotechnology Market Size and Forecast:
-
Market Size in 2025: USD 701.58 billion
-
Market Size by 2035: USD 1,931.91 billion
-
CAGR: 10.66% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get more information on Red Biotechnology Market - Request Free Sample Report
Red Biotechnology Market Trends
-
Rapid adoption of monoclonal antibody-based therapeutics and biosimilars is broadening patient access while intensifying competition among established biologics manufacturers and emerging biotech firms.
-
Expansion of CAR-T cell therapy and next-generation gene therapy pipelines is reshaping oncology treatment paradigms, with over 2,000 active gene and cell therapy clinical trials recorded globally as of 2024.
-
Integration of AI and machine learning platforms into target identification, protein structure prediction, and clinical trial optimization is reducing average drug development timelines by an estimated 20-30%.
-
Growth of contract development and manufacturing organizations (CDMOs) and contract research organizations (CROs) is enabling smaller biotech firms to outsource biologic production, lowering barriers to market entry.
-
Rising regulatory support, including the FDA Breakthrough Therapy Designation and the EMA PRIority MEdicines (PRIME) scheme, is accelerating approval pathways for advanced therapy medicinal products (ATMPs).
-
Personalized medicine approaches driven by companion diagnostics and molecular biomarker identification are increasing the clinical success rates of targeted biologic therapies across oncology, autoimmune, and rare disease indications.
-
Increasing public and private investment in mRNA technology platforms beyond infectious disease vaccines is expanding into oncology, cardiovascular, and metabolic disease applications, generating new revenue streams across the red biotechnology value chain.
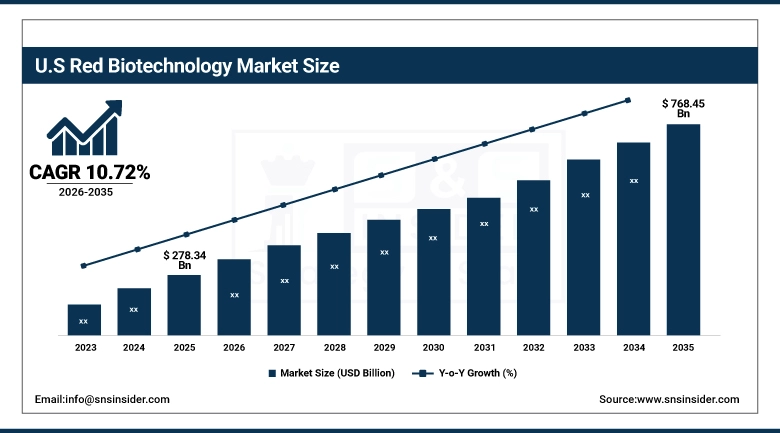
The U.S. Red Biotechnology Market was valued at USD 278.34 billion in 2025 and is expected to reach USD 768.45 billion by 2035, growing at a CAGR of 10.72% from 2026-2035. The United States commands the dominant position in the global red biotechnology landscape, supported by a robust biopharmaceutical manufacturing base, significant NIH and private-sector R&D funding exceeding USD 95 billion annually, and a well-established regulatory environment under the FDA's Center for Biologics Evaluation and Research (CBER) and Center for Drug Evaluation and Research (CDER). The 21st Century Cures Act, Biosimilar Action Plan, and ongoing Inflation Reduction Act provisions continue to shape drug pricing and market access dynamics, while the concentration of leading biotech clusters in Boston, San Francisco, and San Diego sustains a highly innovative and competitive domestic industry ecosystem.
Red Biotechnology Market Growth Drivers:
-
Rising Global Burden of Chronic and Rare Diseases is Driving the Red Biotechnology Market Growth
The increasing global incidence of cancer, autoimmune diseases, cardiovascular diseases, and rare genetic diseases is another major structural driver of growth for the global red biotechnology market. According to the WHO, cancer resulted in 9.7 million deaths in 2023. Biologic drugs currently account for more than 40% of the overall global pharmaceutical revenue, and this is expected to rise even more as biologic drugs progress through their product development pipeline and biosimilar competition continues to erode pharmaceutical costs. The increasing awareness of rare diseases, coupled with government incentives for orphan drugs, is encouraging companies in the global red biotechnology industry to enter new, albeit expensive, areas of research, thus increasing revenue for the overall industry.
For instance, in September 2024, the global biopharmaceutical pipeline comprised more than 7,200 molecules in active clinical development, of which approximately 52% were biologics or biologic-derived compounds, reflecting the sector's dominant role in next-generation drug discovery and development.
Red Biotechnology Market Restraints:
-
High Development Costs and Manufacturing Complexity are Hampering the Red Biotechnology Market Growth
The expensive nature of biologic drug development, which ranges from USD 1.2 billion to USD 2.6 billion per approved molecule, along with the inherent complexity of upstream and downstream bioprocesses, is a major inhibitor of growth for this industry. This is because biologic drugs are not as easy to manufacture as small molecule-based pharmaceuticals, considering their need for cold chain logistics, sterile fill finish, and quality control. This is particularly problematic for smaller and mid-sized red biotechnology companies, who are not able to fund their candidates through late-stage clinical trials. Furthermore, biosimilar price pressure and increasing payer pressure on biologic drug reimbursements are squeezing traditional product franchises.
Red Biotechnology Market Opportunities:
-
Next-Generation Gene and Cell Therapy Platforms are Creating High-Value Growth Opportunities for the Red Biotechnology Market
The commercialization of gene therapy and cell therapy platforms such as in vivo gene editing, allogeneic CAR-T therapy, and viral vector-based systems such as lentiviral and adeno-associated viral vectors is resulting in a high and increasing long-term value creation opportunity within the red biotechnology market. Improvements in large-scale viral vector manufacturing, lower immunogenicity profiles, and robust clinical efficacy results from approved products are helping to build payer confidence and reimbursement schemes within the US, EU, and Japan. The intersection of CRISPR-based gene correction technologies and emerging platforms such as next-generation sequencing and single-cell analysis tools is providing a new and expanding opportunity for one-time curative treatments of diseases such as hemoglobinopathies, inherited metabolic diseases, and solid tumor oncology, representing a market opportunity of over $130 billion by 2035.
For instance, in February 2025, global investment in cell and gene therapy development surpassed USD 18.4 billion across 320 active funding rounds, signaling strong investor confidence in the commercial scalability of advanced therapeutic platforms within the red biotechnology ecosystem.
Red Biotechnology Market Segment Analysis
-
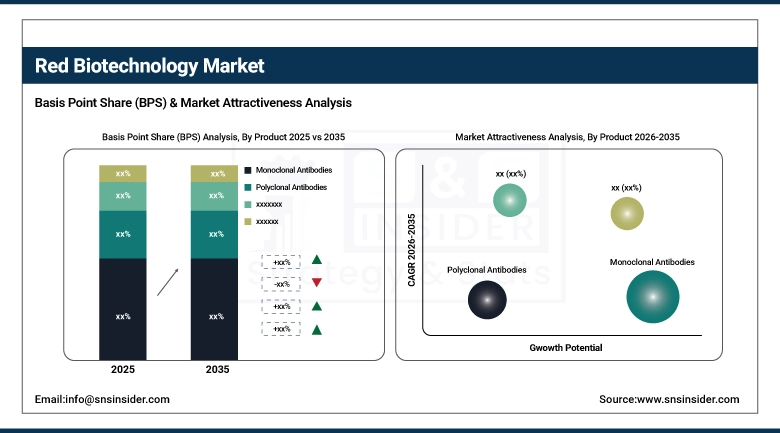
By product, monoclonal antibodies held the largest share of approximately 36.42% in 2025, and the gene therapy products segment is expected to register the highest growth with a CAGR of 14.82%.
-
By end-user, pharmaceutical and biotechnology companies accounted for the leading share of nearly 58.76% in 2025, while the CMOs and CROs segment is expected to register the highest growth with a CAGR of 12.34%.
By Product, Monoclonal Antibodies Lead the Market, While Gene Therapy Products Register Fastest Growth
The monoclonal antibodies segment has continued to hold the highest market revenue share of 36.42% in 2025, owing to the commercial success of monoclonal antibodies in oncology and autoimmune diseases, clinical validation of over 100 approved monoclonal antibodies, and ongoing lifecycle management activities for monoclonal antibodies by top biopharma companies. The revenue growth rate for this segment is also being driven by the accelerated development of monoclonal antibodies, such as bispecific antibodies and ADCs. On the other hand, the gene therapy products segment is projected to witness the highest CAGR of 14.82% over the forecast period of 2026-2035, owing to increased FDA and EMA approvals, improved AAV and lentiviral vector manufacturing capabilities, and clinical validation of the long-term efficacy of gene therapies for the treatment of rare and orphan diseases.
By End-User, Pharmaceutical and Biotechnology Companies Lead and Maintain Strong Growth Momentum
Pharmaceutical and biotechnology companies held the largest share of 58.76% as the end user segment in 2025, owing to their vertical integration capabilities from R&D to commercialization, large-scale biologics manufacturing capabilities, and dedicated biologic drug portfolios generating consistent revenue for the companies. Increasing biologics licensing agreements and the development pipeline for biosimilars are further contributing to the market share held by this segment. The CMOs and CROs segment is expected to witness the highest CAGR of 12.34% during the forecast period, indicating a long-term trend for the outsourced biologic drug manufacturing and development market from both the large pharma and biotech sectors.
Red Biotechnology Market Regional Highlights:
North America Red Biotechnology Market Insights:
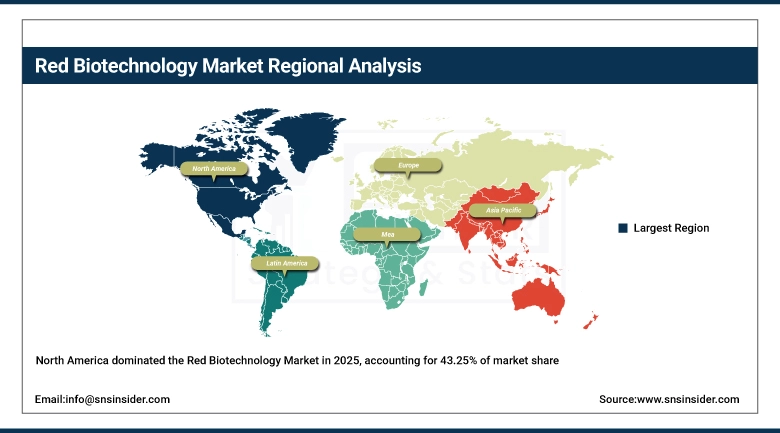
North America held the largest revenue share of approximately 43.25% in 2025 in the global red biotechnology market, supported by the highest concentration of biopharmaceutical companies, biotechnology startups, and academic research institutions globally. The United States alone accounts for over 39% of total global biologic drug sales, anchored by a mature payer environment, high biologics reimbursement rates, and FDA regulatory efficiency for novel biologic approvals. Sustained NIH funding levels, active venture capital ecosystems supporting early-stage biotech ventures, and strong intellectual property protections continue to reinforce North America's market leadership position throughout the forecast period.
Get Customized Report as per Your Business Requirement - Enquiry Now
Asia Pacific Red Biotechnology Market Insights:
Asia Pacific is the highest growth region for the global red biotechnology industry, growing at a CAGR of 12.84%, owing to the expansion of biopharmaceutical production facilities in countries like China, India, South Korea, and Japan, along with government-led initiatives for a bio-based economy and increasing healthcare spending in these countries. The Chinese government's national plan for biotechnology development under the 14th Five-Year Plan, India's Biotechnology Industry Research Assistance Council programs, and South Korea's bio-clustering strategy based on the Songdo International Bio District are all set to attract multi-billion-dollar investments in red biotechnology. Biosimilar exports from India, biologic drug approvals in China, and academic collaboration pipelines are all set to make Asia Pacific a key growth driver for global red biotechnology revenues until 2035.
Europe Red Biotechnology Market Insights:
Europe, as the second-largest biotech market, has been driven by high biosimilar uptake rates, well-developed regulatory standards via the EMA, and well-funded healthcare systems that enable widespread access to biologic therapies. The region also has the advantage of a well-developed bioclustering industry, particularly in Germany, Switzerland, the UK, Denmark, and Belgium, where there are significant spillovers of international collaborations in the life sciences sector, as well as the EU’s Horizon Europe funding initiative. High biosimilar uptake rates of over 60% for certain biologic therapies in the EU have driven the region’s forward-looking strategy to deliver cost-effective access to biologic therapies, thereby creating a consistent demand for next-generation biologic production technologies.
Latin America (LATAM) and Middle East & Africa (MEA) Red Biotechnology Market Insights:
In Latin America and Middle East & Africa, the increasing investments in healthcare, the rise of pharmaceutical manufacturing capabilities, and the increasing availability of biologic therapies through government healthcare programs are contributing to the steady growth of the red biotechnology market. The increasing biologic drug regulations in Brazil and the healthcare revolution plans for Saudi Arabia under Vision 2030 are boosting investments in local biopharmaceutical manufacturing and R&D capabilities. The increasing partnerships between multinational red biotechnology companies and healthcare institutions are boosting the growth of the red biotechnology market in both regions.
Red Biotechnology Market Competitive Landscape:
Roche Holdings AG (est. 1896) is a global pioneer in biopharmaceuticals and diagnostics, commanding a leading position in the red biotechnology market through its oncology biologics portfolio, HER2-targeted therapies, and next-generation antibody engineering capabilities across multiple high-value therapeutic areas.
-
In January 2025, Roche announced a USD 1.1 billion expansion of its biologics manufacturing facility in Basel, Switzerland, targeting increased large-scale antibody production capacity to meet growing demand for its oncology and immunology biologic pipeline.
Amgen Inc. (est. 1980) is a leading independent biopharmaceutical company and a foundational red biotechnology innovator, with a diversified portfolio spanning recombinant proteins, monoclonal antibodies, and biosimilars, supported by industry-leading bioprocessing and cell line development infrastructure.
-
In March 2025, Amgen initiated Phase III clinical trials for its next-generation bispecific antibody targeting solid tumors in NSCLC, reinforcing its oncology pipeline investment and expanding its competitive position in the high-growth antibody engineering segment.
Moderna, Inc. (est. 2010) is a transformative red biotechnology company pioneering the clinical application of mRNA-based therapeutics and vaccines beyond infectious diseases, with active development programs in oncology, cardiovascular disease, rare metabolic disorders, and autoimmune indications leveraging its proprietary lipid nanoparticle delivery platform.
-
In November 2024, Moderna reported positive Phase II data for its personalized mRNA cancer vaccine in combination with pembrolizumab, demonstrating a 44% reduction in recurrence risk for high-risk melanoma patients and validating the commercial potential of individualized biologic cancer immunotherapy.
Red Biotechnology Market Key Players:
-
Roche Holdings AG
-
Johnson & Johnson (Janssen Pharmaceuticals)
-
Pfizer Inc.
-
Novartis AG
-
AbbVie Inc.
-
Bristol-Myers Squibb Company
-
Amgen Inc.
-
Merck & Co., Inc.
-
AstraZeneca PLC
-
Gilead Sciences, Inc.
-
Regeneron Pharmaceuticals, Inc.
-
Biogen Inc.
-
Moderna, Inc.
-
Sanofi S.A.
-
Eli Lilly and Company
-
GSK plc
-
Bayer AG
-
Novo Nordisk A/S
-
CSL Behring LLC
-
Lonza Group AG
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 701.58 Billion |
| Market Size by 2035 | USD 1,931.91 Billion |
| CAGR | CAGR of 10.66% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Product (Monoclonal Antibodies, Polyclonal Antibodies, Recombinant Proteins, Vaccines, Cell-Based Immunotherapy Products, Gene Therapy Products, Cell Therapy Products, Tissue-Engineered Products, Stem Cells, Cell Culture, Viral Vector, Enzymes, Kits And Reagents, Animal Models, Molecular Diagnostics, and Others) • By End-user (Academic Research Institutes, CMOs & CROs, Pharmaceutical & Biotechnology Companies, and Others) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | Roche Holdings AG, Johnson & Johnson (Janssen Pharmaceuticals), Pfizer Inc., Novartis AG, AbbVie Inc., Bristol-Myers Squibb Company, Amgen Inc., Merck & Co., Inc., AstraZeneca PLC, Gilead Sciences, Inc., Regeneron Pharmaceuticals, Inc., Biogen Inc., Moderna, Inc., Sanofi S.A., Eli Lilly and Company, GSK plc, Bayer AG, Novo Nordisk A/S, CSL Behring LLC, Lonza Group AG |

