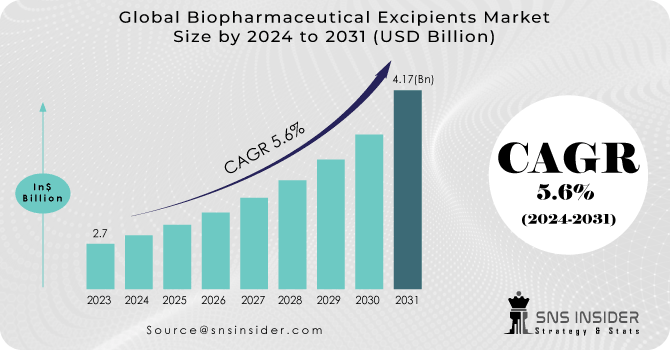
The Biopharmaceutical Excipients Market Size was valued at USD 2.7 billion in 2023 and is expected to reach USD 4.17 billion by 2031, and grow at a CAGR of 5.6% over the forecast period 2024-2031.
Increasing R&D investments in biosimilars, coupled with the patent expiration of top-selling biologics, are projected to propel the market. The chemical and physical instability of biopharmaceuticals and stringent manufacturing requirements need to be fulfilled for consistent yield of biologics and to ensure high cell viability are boosting the market. Furthermore, a growing adoption of multifunctional excipients, expiry of bioactive patents and increasing focus by pharmaceutical manufacturers on the Generic Market. In addition, to preserve the stability of medicinal products, new biotechnological excipients are being developed by operational players. In addition, Roquette launched the new excipient PEARLITOL ProTec in Europe in May 2023. It is a unique, plant based, processed blend of maize starch and mannitol, which can be used for the stabilisation of probiotics and for the protection of sensitive active ingredients such as mannitol.
Get more information on Biopharmaceutical Excipients Market - Request Sample Report
Biopharmaceutical excipients, which are inactive compounds used as carriers, must be coupled with the active pharmaceutical ingredients in order to achieve an appropriate formulation that is able to prolong its shelf life. The stability of the product must be improved by careful use of a combination of excipients, given that biopharma molecules are prone to instability.
DRIVERS
The rapid development of the legal environment for production of biosimilars
Increasing prevalence of cancer and infectious diseases
As cancer incidence increases, the need for diverse biological treatment is increasing. Biopharma companies, both large and small, are still looking for the molecular causes of cancer as well as developing drugs to stop tumor cells from growing. A number of aspects of the oncology market have therefore been shaped by the biopharma industry. Moreover, there is a significant burden of communicable diseases in industrialized and developing countries. Especially in low- and middle-income nations, contagious diseases, including HIV/AIDS, TB, malaria, and many others, cause a high mortality rate.
RESTRAINTS
The market growth rate will slow down because of the lack of common treatments
Regulatory issues in the manufacture of biopharmaceutical ingredients
The quality and safety of medicinal products is one of the most important issues for regulators. Regulators have become more stringent in their stance against the adulteration of pharmaceuticals with excipients and APIs due to a number of strict measures adopted last few years. However, there is a need for an independent excipient approval pathway to limit the entry of new excipients and new drugs into the market in the current regulatory environment. Moreover, due to uncertainty as to whether the new excipient will be accepted into a regulatory review which hinders market growth, manufacturers and producers of products are hesitant to invest significant resources and time in its development.
OPPORTUNITIES
A growing demand for novel excipients in the development of medicines
New excipients are essential to ensure that new, better and safer drugs can be developed for the biopharma market. The majority of approved products are made up of conventional, well studied compendial excipients; therefore, the use of novel excipients is not necessary to formulate certain advanced products. Moreover, more companies are choosing to use sophisticated and innovative formulations containing new ingredients. A novel excipient is defined by the FDA as an excipient that has not been used in an FDA approved medicinal product. Therefore, there is no indication of any use in food.
CHALLENGES
Lack of awareness of the biopharmaceutical excipients
Many of the Ukraine-based clinical trial sites have collapsed, as the Russian invasion has destroyed hospitals and research facilities. In Russia, biopharma companies are stopping enrolling in a number of clinical studies. Several pharmaceutical and biotechnology companies acknowledged the likelihood of delays in trials in Ukraine and Russia, as well as difficulties to produce vaccines and medicinal products that are required for these countries. The instability in the region may lead to currency depreciation, trade tariffs and export restrictions that could have a negative impact on biopharma excipient prices from affected regions. This uncertainty could lead to disruption of market dynamics and pricing strategies for pharmaceuticals, which would have a negative impact on their bottom lines as well as the possible increase in consumer costs. In addition, the approval and certification of pharmaceutical products and excipients by disrupting regulatory processes and controls. As a result, companies may seek alternative sources of supply which could lead to changes in market dynamics and competition due to the resulting uncertainty regarding compliance with regulatory requirements.
During economic slowdowns, excipient manufacturers and suppliers may encounter difficulties accessing financing, which could hamper their capacity expansion, technology upgrades, and product development efforts. Consequently, this may result in supply chain constraints and potential shortages of essential excipients in the market. In addition, during economic downturns, regulatory agencies may face budgetary constraints or prioritise the use of resources to address immediate public health issues in order to speed up approval processes for new excipients or updates affecting existing excipients.
In spite of these challenges, the market for biotherapeutic excipients may also present opportunities such as to reduce costs, pharmaceutical companies may be more focused on the development of generic or biogeneric versions of currently available biopharmaceuticals and this could lead to an increase in excipient demand from such formulations.
Market segmentation
By Product
Solubilizes & Surfactants/Emulsifiers
Triglycerides
Esters
Others
Polyols
Mannitol
Sorbitol
Others
Carbohydrates
Sucrose
Dextrose
Starch
Others
In 2023, carbohydrates accounted for 36.2% of the market's revenue and this trend is expected to be maintained over the forecast period. In particular, the dominant share of this segment can be attributed to the extensive use of starch, sucrose and dextrose in different medicinal products. The rapid adoption of high-purity carbohydrates like trehalose in biosimilar development is driven by their capacity to boost final product yield, stabilize biologics, prevent aggregation, and enhance the overall biopharmaceutical manufacturing process. Additionally, the growing acceptance of carbohydrates as bulking agents for lyophilizing biomolecules, such as proteins, further contributes to segment growth.
The polyol segment is anticipated to grow at a faster compound annual growth rate over the forecast period. Polyols are sugar alcohols in some fruits, vegetables and sugars free sweeteners. The growing demand for protein therapeutic products is a reason for the growth in this segment. In order to enhance the tonic and stability of proteins, phenols are added. In addition, they have been shown to provide greater thermodynamic stability, which is essential for protein-based formulations.
In 2023, North America had the largest market share at 32.3%. Several large pharmaceutical manufacturing companies operating in this region are likely to be responsible for the growth. In addition, there has been a significant demand for Biopharmaceutical production in this region and the need to maintain high quality raw materials used as development ingredients. Therefore, the use of biopharma excipients that comply with regulatory standards has been required by a number of regional excipient manufacturers in order to achieve the necessary production scale. Moreover, new studies are being carried out by companies active in the region to develop efficient excipient solutions.
Asia Pacific market is expected to grow at a faster rate of around 6 % during the forecast period due to the presence of major CMOs involved in drug development. The development of biosimilars is largely driven by countries in this region, such as South Korea, India, and China. There are many reasons for the widespread development of biosimilars, including their large patient base, cost advantages, affordability and favourable legal framework. There are many biopharma companies active in Asian countries that could be potential customers on the market. As a result of the substantial cost advantages provided by the value chain, international biopharma companies are involved in relocating their manufacturing and research and development operations to these countries. In turn, the demand for novel excipients is increasing as a result of the increasing demand for biosimilars.
REGIONAL COVERAGE:
North America
US
Canada
Mexico
Europe
Eastern Europe
Poland
Romania
Hungary
Turkey
Rest of Eastern Europe
Western Europe
Germany
France
UK
Italy
Spain
Netherlands
Switzerland
Austria
Rest of Western Europe
Asia Pacific
China
India
Japan
South Korea
Vietnam
Singapore
Australia
Rest of Asia Pacific
Middle East & Africa
Middle East
UAE
Egypt
Saudi Arabia
Qatar
Rest of the Middle East
Africa
Nigeria
South Africa
Rest of Africa
Latin America
Brazil
Argentina
Colombia
Rest of Latin America
Some of the major key players are Associated British Foods, BASF SE, Colorcon, Clariant, DFE Pharma, Evonik Industries AG, IMCD, Sigachi Industries Pvt. Ltd., Merck KGaA, J. RETTENMAIER & SOHNE GmbH + Co KG, Roquette Freres, Signet Chemical Corporation Pvt, Ltd. and Other Players.
In December 2022- BASF SE and StePac Ltd have collaborated to design the next generation of environmentally friendly packaging for the fresh produce sector. Utilizing BASF's delivery of Ultramid Cycled TM, a chemically recycled polyamide 6, StePac aims to enhance contact-sensitive package designs to meet more sustainable standards within the circular economy.
In September 2022- Spectrum Chemical Mfg. Corp., renowned for its provision of specialty, fine, and cGMP chemicals, along with laboratory equipment and supplies worldwide, has established a new U.S. distribution partnership with DFE Pharma, a prominent global provider of excipient solutions for pharmaceutical and nutraceutical industries.
| Report Attributes | Details |
|---|---|
| Market Size in 2023 | US$ 2.7 Billion |
| Market Size by 2031 | US$ 4.17 Billion |
| CAGR | CAGR of 5.6% From 2024 to 2031 |
| Base Year | 2023 |
| Forecast Period | 2024-2031 |
| Historical Data | 2020-2022 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Product (Solubilizes & Surfactants/Emulsifiers (Triglycerides, Esters, Others), Polyols (Mannito, Sorbitol, Others), Carbohydrates (Sucrose, Dextrose, Starch, Others), By Regions and Global Forecast 2024-2031 |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]), Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia, Rest of Latin America) |
| Company Profiles | Associated British Foods, BASF SE, Colorcon, Clariant, DFE Pharma, Evonik Industries AG, IMCD, Sigachi Industries Pvt. Ltd., Merck KGaA, J. RETTENMAIER & SOHNE GmbH + Co KG, Roquette Freres, Signet Chemical Corporation Pvt, Ltd. |
| Key Drivers | • Significantly increasing need for big molecule development • Rapid expansion of the legal environment for the creation of biosimilars |
| RESTRAINTS | • The lack of common treatments will slow the market's rate of expansion. |
Ans: The Biopharmaceutical Excipients Market Size was valued at USD 2.7 billion in 2023
Ans. The Biopharmaceutical Excipients Market is to grow at a CAGR of 5.6% over the forecast period 2024-2031
Ans. The greatest market share was held by North America.
Ans. Associated British Foods, BASF SE, Colorcon, Clariant, DFE Pharma, Evonik Industries AG, IMCD, Sigachi Industries Pvt. Ltd are the key players of Biopharmaceutical Excipients market
Ans. Lack of awareness is the challenges faced by Biopharmaceutical Excipients market.
Table of Contents
1. Introduction
1.1 Market Definition
1.2 Scope
1.3 Research Assumptions
2. Research Methodology
3. Market Dynamics
3.1 Drivers
3.2 Restraints
3.3 Opportunities
3.4 Challenges
4. Impact Analysis
4.1 COVID-19 Impact Analysis
4.2 Impact of Ukraine- Russia War
4.3 Impact of Ongoing Recession
4.3.1 Introduction
4.3.2 Impact on major economies
4.3.2.1 US
4.3.2.2 Canada
4.3.2.3 Germany
4.3.2.4 France
4.3.2.5 United Kingdom
4.3.2.6 China
4.3.2.7 Japan
4.3.2.8 South Korea
4.3.2.9 Rest of the World
5. Value Chain Analysis
6. Porter’s 5 forces model
7. PEST Analysis
8. Biopharmaceutical Excipients Market Segmentation, By Product type
8.1 Solubilizers & Surfactants/Emulsifiers
8.1.1 Triglycerides
8.1.2 Esters
8.2 Polyols
8.2.1 Mannitol
8.2.2 Sorbitol
8.3 Carbohydrates
8.3.1 Sucrose
8.3.2 Dextrose
8.3.3 Starch
8.4 Specialty Excipients
9. Regional Analysis
9.1 Introduction
9.2 North America
9.2.1 USA
9.2.2 Canada
9.2.3 Mexico
9.3 Europe
9.3.1 Germany
9.3.2 UK
9.3.3 France
9.3.4 Italy
9.3.5 Spain
9.3.6 The Netherlands
9.3.7 Rest of Europe
9.4 Asia-Pacific
9.4.1 Japan
9.4.2 South Korea
9.4.3 China
9.4.4 India
9.4.5 Australia
9.4.6 Rest of Asia-Pacific
9.5 The Middle East & Africa
9.5.1 Israel
9.5.2 UAE
9.5.3 South Africa
9.5.4 Rest
9.6 Latin America
9.6.1 Brazil
9.6.2 Argentina
9.6.3 Rest of Latin America
10. Company Profiles
10.1 Associated British Foods
10.1.1 Financial
10.1.2 Products/ Services Offered
10.1.3 SWOT Analysis
10.1.4 The SNS view
10.2 BASF SE
10.3 Colorcon
10.4 Clariant
10.5 DFE Pharma
10.6 Evonik Industries AG
10.7 IMCD
10.8 Sigachi Industries Pvt. Ltd.
10.9 Merck KGaA
10.10 J. RETTENMAIER & SOHNE GmbH + Co KG
11. Competitive Landscape
11.1 Competitive Benchmark
11.2 Market Share Analysis
11.3 Recent Developments
12. Conclusion
An accurate research report requires proper strategizing as well as implementation. There are multiple factors involved in the completion of good and accurate research report and selecting the best methodology to compete the research is the toughest part. Since the research reports we provide play a crucial role in any company’s decision-making process, therefore we at SNS Insider always believe that we should choose the best method which gives us results closer to reality. This allows us to reach at a stage wherein we can provide our clients best and accurate investment to output ratio.
Each report that we prepare takes a timeframe of 350-400 business hours for production. Starting from the selection of titles through a couple of in-depth brain storming session to the final QC process before uploading our titles on our website we dedicate around 350 working hours. The titles are selected based on their current market cap and the foreseen CAGR and growth.
The 5 steps process:
Step 1: Secondary Research:
Secondary Research or Desk Research is as the name suggests is a research process wherein, we collect data through the readily available information. In this process we use various paid and unpaid databases which our team has access to and gather data through the same. This includes examining of listed companies’ annual reports, Journals, SEC filling etc. Apart from this our team has access to various associations across the globe across different industries. Lastly, we have exchange relationships with various university as well as individual libraries.
Step 2: Primary Research
When we talk about primary research, it is a type of study in which the researchers collect relevant data samples directly, rather than relying on previously collected data. This type of research is focused on gaining content specific facts that can be sued to solve specific problems. Since the collected data is fresh and first hand therefore it makes the study more accurate and genuine.
We at SNS Insider have divided Primary Research into 2 parts.
Part 1 wherein we interview the KOLs of major players as well as the upcoming ones across various geographic regions. This allows us to have their view over the market scenario and acts as an important tool to come closer to the accurate market numbers. As many as 45 paid and unpaid primary interviews are taken from both the demand and supply side of the industry to make sure we land at an accurate judgement and analysis of the market.
This step involves the triangulation of data wherein our team analyses the interview transcripts, online survey responses and observation of on filed participants. The below mentioned chart should give a better understanding of the part 1 of the primary interview.
Part 2: In this part of primary research the data collected via secondary research and the part 1 of the primary research is validated with the interviews from individual consultants and subject matter experts.
Consultants are those set of people who have at least 12 years of experience and expertise within the industry whereas Subject Matter Experts are those with at least 15 years of experience behind their back within the same space. The data with the help of two main processes i.e., FGDs (Focused Group Discussions) and IDs (Individual Discussions). This gives us a 3rd party nonbiased primary view of the market scenario making it a more dependable one while collation of the data pointers.
Step 3: Data Bank Validation
Once all the information is collected via primary and secondary sources, we run that information for data validation. At our intelligence centre our research heads track a lot of information related to the market which includes the quarterly reports, the daily stock prices, and other relevant information. Our data bank server gets updated every fortnight and that is how the information which we collected using our primary and secondary information is revalidated in real time.

Step 4: QA/QC Process
After all the data collection and validation our team does a final level of quality check and quality assurance to get rid of any unwanted or undesired mistakes. This might include but not limited to getting rid of the any typos, duplication of numbers or missing of any important information. The people involved in this process include technical content writers, research heads and graphics people. Once this process is completed the title gets uploader on our platform for our clients to read it.
Step 5: Final QC/QA Process:
This is the last process and comes when the client has ordered the study. In this process a final QA/QC is done before the study is emailed to the client. Since we believe in giving our clients a good experience of our research studies, therefore, to make sure that we do not lack at our end in any way humanly possible we do a final round of quality check and then dispatch the study to the client.
The Conjunctivitis Treatment Market size was USD 4.2 Billion in 2023 and is expected to Reach USD 6.06 Billion by 2031 and grow at a CAGR of 4.7% over the forecast period of 2024-2031.
The Surgical Procedures Market size was estimated USD 10,827 thousands in 2022 and is expected to reach USD 14257.09 thousands by 2030 at a CAGR of 3.5% during the forecast period of 2023-2030.
Laboratory Freezers Market Report Scope & Overview: Laboratory Freezers Market Size was valued at
The Urinary Drainage Bags Market size was valued at USD 2.71 billion in 2023 and it is expected to be worth around USD 3.75 billion by 2031 growing at a remarkable GAGR of 4.86% over the forecast period 2024-2031.
The Intensive Care Unit (ICU) Market size was valued at $ 3.10 Bn in 2022 and is expected to reach $ 9.45 Bn by 2030,and grow at CAGR of 14.5 % over the forecast period of 2023-2030.
In 2022, The Skin Cancer Treatment Market size amounted to USD 9.98 Billion & is estimated to reach USD 18.06 Billion by 2030 and increase at a compound annual growth rate of 7.7% between 2023 and 2030.
Hi! Click one of our member below to chat on Phone