Pharmacovigilance Market Report Scope and Overview:
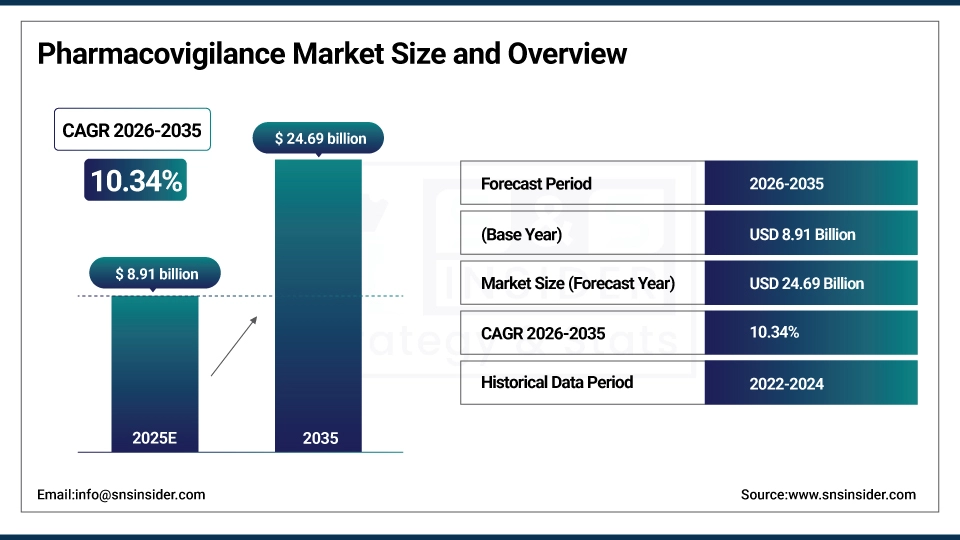
The Pharmacovigilance Market size is estimated at USD 8.91 billion in 2025 and is expected to reach USD 24.69 billion by 2035, growing at a CAGR of 10.34% over the forecast period of 2026-2035.
The global pharmacovigilance market trend is a growing demand for drug safety monitoring solutions such as adverse event reporting systems, signal detection platforms, and risk management services as the growth of the market is driven by increasing regulatory requirements for post-market surveillance, rising adverse drug reaction incidents, and pharmaceutical industry expansion into emerging markets.
For instance, in March 2024, growing awareness and improved regulatory compliance drove a 26% increase in pharmacovigilance service adoption for pharmaceutical companies globally, boosting drug safety monitoring and adverse event reporting capabilities.
Pharmacovigilance Market Size and Forecast:
-
Market Size in 2025: USD 8.91 billion
-
Market Size by 2035: USD 24.69 billion
-
CAGR: 10.34% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
Get More Information on Pharmacovigilance Market - Request Free Sample Report
Pharmacovigilance Market Trends
-
Pharmacovigilance solutions are being adopted because pharmaceutical companies demand robust adverse event monitoring, regulatory compliance management, and real-time safety signal detection.
-
Customized drug safety monitoring tools based on therapeutic areas, patient demographics, and geographic regulations to improve patient safety outcomes.
-
The development of AI-powered adverse event detection, machine learning algorithms, and natural language processing to improve the accuracy of safety monitoring and reduce manual case processing burden.
-
Automated case intake systems, electronic health record integration, and real-time data analytics are all available to ensure continuous safety surveillance and regulatory reporting compliance.
-
Increased demand for cloud-based pharmacovigilance platforms, mobile reporting applications and centralized safety databases to help global data accessibility and regulatory submission.
-
Collaboration between pharmaceutical manufacturers, contract research organizations and regulatory authorities to develop standardized safety reporting systems and improve data interoperability.
-
FDA, EMA and WHO promoting standards for adverse event reporting, ICH E2B compliance, periodic safety update reports, and risk management plan requirements.
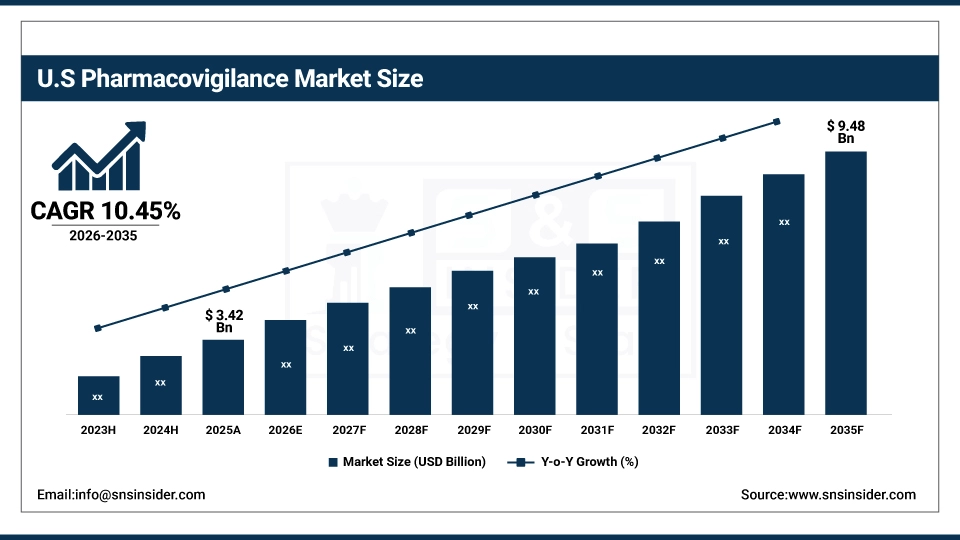
The U.S. Pharmacovigilance Market is projected to be USD 3.42 billion in 2025 and is expected to reach USD 9.48 billion in 2035, growing at a CAGR of 10.45% from 2026 to 2035. The United States is the largest market for pharmacovigilance. This is mainly due to the strict FDA regulations, the high level of pharmaceutical R&D activities, and the established drug safety surveillance system. Government mandates, high levels of adverse event reporting, and increased investment in patient safety programs from pharmaceutical companies help to drive growth in the market.
Pharmacovigilance Market Growth Drivers:
-
Stringent Regulatory Requirements and Compliance Mandates are Driving the Pharmacovigilance Market Growth
Stringent regulatory requirements and compliance mandates take the center stage as a growth driver for the pharmacovigilance market share, and are driven by the implementation of FDA adverse event reporting systems, EMA pharmacovigilance guidelines, and WHO safety monitoring standards for increased drug safety surveillance and post-market monitoring. These solutions for pharmaceutical safety management and risk mitigation are driving the base of the market, the penetration of contract outsourcing & in-house markets, and adding to the overall market share globally.
For instance, in June 2024, automated and cloud-based pharmacovigilance solutions accounted for ~62% of the total global pharmaceutical safety technology investments, reflecting growing institutional preference and expanding market share.
Pharmacovigilance Market Restraints:
-
High Implementation Costs and Resource Constraints are Hampering the Pharmacovigilance Market Growth
High implementation costs & resource constraints of pharmacovigilance systems also restrict the pharmacovigilance market growth, as a large number of small and medium-sized pharmaceutical companies face difficulties establishing comprehensive safety monitoring infrastructure and maintaining qualified pharmacovigilance professionals. This might lead to underreporting, limited system adoption, and reduced efficiency for drug safety organizations. As a result, patient safety outcomes suffer, and market growth is stunted in regions where pharmaceutical companies have limited budgets and pharmacovigilance expertise is scarce.
Pharmacovigilance Market Opportunities:
-
AI Integration and Advanced Analytics Drive Future Growth Opportunities for the Pharmacovigilance Market
The opportunity in the AI integration and advanced analytics in pharmacovigilance market is in the form of predictive safety signal detection, automated case processing, and intelligent adverse event pattern recognition. These solutions provide for early safety concern identification, individualized risk assessment, and real-time pharmacovigilance data mining. Through enhanced regulatory compliance, clinical trial safety monitoring, and operational efficiency, particularly in areas with complex drug development pipelines, these technologies may improve patient outcomes, decrease drug withdrawal incidents, and expand the market.
For instance, in April 2024, the FDA reported that 68% of major pharmaceutical companies implemented AI-driven pharmacovigilance platforms, highlighting rising technology adoption and increasing demand for intelligent drug safety monitoring tools.
Pharmacovigilance Market Segment Analysis
-
By product life cycle, Phase IV held the largest share of around 38.64% in 2025, and the Phase III segment is expected to register the highest growth with a CAGR of 11.12%.
-
By service provider, the contract outsourcing segment dominated the market with approximately 61.23% share in 2025, while the in-house is expected to register the highest growth with a CAGR of 10.87%.
-
By end use, pharmaceuticals accounted for the leading share of nearly 52.34% in 2025, and is expected to register the highest growth with a CAGR of 10.56%.
By Product Life Cycle, Phase IV dominates the market, while Phase III experiences the fastest growth
The Phase IV market is expected to hold the largest revenue share of about 38.64% in 2025, due to the extensive post-market surveillance needs, long-term safety monitoring obligations, and exposure of a large patient population to commercialized drugs. Emerging trends include the growing regulatory interest in the collection of real-world evidence and the mandatory periodic safety update reports.
The Phase III market is expected to register the fastest growth at a CAGR of nearly 11.12% during the forecast period of 2026-2035, due to the growing number of late-stage clinical trials, the need for stringent safety reporting, and the rising investment in drug development initiatives. Factors include the rising complexity of clinical trial designs, the desire for proactive safety monitoring in pivotal trials.
By Service Provider, the Contract Outsourcing Segment dominates, while the In-House Segment Shows Rapid Growth
By 2025, the contract outsourcing segment contributed the largest revenue share of 61.23% due to cost-effectiveness, access to specialized expertise and scalability advantages for pharmaceutical companies. Growing adoption of contract research organization services coupled with resource optimization strategies, companies are increasingly outsourcing pharmacovigilance activities.
The in-house segment is projected to grow at the highest CAGR of about 10.87% between 2026 and 2035 due to the growing need for greater control over safety data and direct regulatory interaction capabilities. Some of the reasons include enhanced data security concerns, better integration with internal clinical development teams, and large pharmaceutical companies' preference for maintaining proprietary safety monitoring systems.
By End Use, Pharmaceuticals Lead, and Registers Fastest Growth
The pharmaceuticals segment accounted for the largest share of the pharmacovigilance market with about 52.34%, owing to their extensive drug portfolios, global regulatory compliance obligations, and significant investment capacity for safety monitoring infrastructure. Reasons driving the pharmaceutical segment include increasing drug development activities and stringent post-market surveillance requirements. In addition, it is slated to grow at the fastest rate with a CAGR of around 10.56% throughout the forecast period of 2026–2035, as pharmaceutical companies seek comprehensive adverse event monitoring platforms, risk management system implementations, and patient safety enhancement capabilities. Increased focus on pharmacovigilance automation and integration with clinical trial management contribute to their adoption, while improved regulatory compliance and reduced drug safety incidents drive continued investment.
Pharmacovigilance Market Regional Highlights:
North America Pharmacovigilance Market Insights:
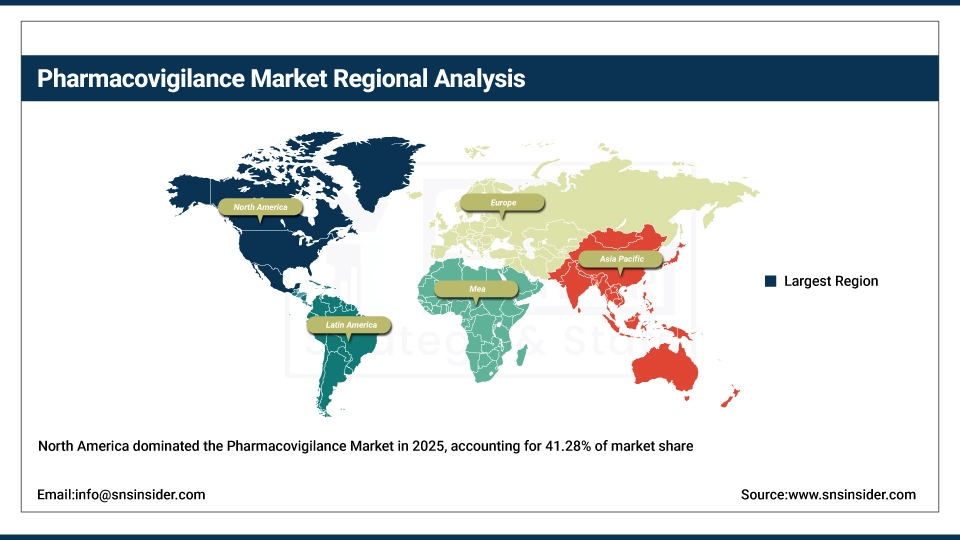
North America held the largest revenue share of over 41.28% in 2025 of the pharmacovigilance market due to an established pharmaceutical industry infrastructure, stringent FDA safety reporting requirements, and increased healthcare provider awareness regarding the importance of adverse event monitoring. Drivers include ubiquitous use of electronic adverse event reporting systems, an advanced healthcare technology environment, growing pharmaceutical research and development investment and greater acceptance of outsourced pharmacovigilance services stemming from cost optimization needs. Various government safety initiatives, FDA Sentinel System implementation and enormous investments in drug safety technology from pharmaceutical manufacturers are anchoring pharmacovigilance software and services in the market, and ensuring multibillion dollar revenues around the world.
Need any customization research on Pharmacovigilance Market - Enquiry Now
Asia Pacific Pharmacovigilance Market Insights:
Asia Pacific is the fastest-growing segment in the pharmacovigilance market with a CAGR of 12.18% in the forecasted period, as the awareness about drug safety monitoring, government regulatory harmonization initiatives, and pharmaceutical manufacturing expansion in developing nations is growing. Factors including rapid clinical trial outsourcing, rising generic drug production with safety monitoring needs, and growing adoption of international pharmacovigilance standards are stimulating the market growth. Contract research organization establishment and biotechnology sector growth have been instrumental in improving safety surveillance capabilities, especially in countries with emerging pharmaceutical industries. Public health programs and regulatory authority strengthening also help in advancing drug safety monitoring and adverse event reporting systems. Increase in demand in Asia Pacific region owing to rising healthcare expenditure against historical spending levels and growing affordability and accessibility of cloud-based pharmacovigilance services.
Europe Pharmacovigilance Market Insights:
In 2025, the pharmacovigilance market in Europe is the second-dominating region after North America on account of an increase in the adoption of EudraVigilance reporting systems, robust pharmaceutical regulatory frameworks including EMA guidelines, and increasing patient safety empowerment initiatives across healthcare systems. Rising implementation of European pharmacovigilance legislation, advanced medical device safety monitoring, favorable government funding for drug safety programs, and cross-border adverse event reporting harmonization are also contributing to the sustained growth of the market in leading European countries.
Latin America (LATAM) and Middle East & Africa (MEA) Pharmacovigilance Market Insights:
In Latin America, and Middle East & Africa, the growing pharmaceutical sector development and increase in regulatory framework strengthening with clinical trial activity expansion support the pharmacovigilance market growth. The rising popularity of affordable outsourced pharmacovigilance services and regional safety monitoring capabilities, along with international pharmaceutical company presence, will aid drug safety compliance and adverse event reporting. The increasing healthcare infrastructure investment and improving regulatory authority capabilities in these regions are continuing to encourage market growth.
Pharmacovigilance Market Competitive Landscape:
IQVIA Holdings Inc. (est. 1982) is a leading pharmaceutical analytics and contract research organization that focuses on integrated healthcare data solutions for clinical development and commercialization. It uses its comprehensive global database and advanced analytics capabilities to produce cutting-edge pharmacovigilance technology with seamless safety monitoring workflow integration.
-
In February 2025, it expanded its pharmacovigilance platform capabilities with AI-driven signal detection and automated case processing, aiming to improve drug safety monitoring efficiency and regulatory compliance across its global pharmaceutical client network.
Accenture plc (est. 1989) is a well-known global professional services company focused on technology consulting, digital transformation, and business process outsourcing. It invests in intelligent pharmacovigilance platforms and cloud-based safety monitoring applications with the hopes of revolutionizing drug safety management with secure, scalable, and AI-enhanced safety surveillance tools.
-
In May 2024, launched an enhanced cloud-native pharmacovigilance solution featuring real-time adverse event analytics and integrated regulatory reporting capabilities across global pharmaceutical markets, enhancing safety data management, compliance automation, and signal detection accuracy.
Oracle Corporation (founded in 1977) is a major enterprise software and cloud solutions company in the areas of database management, healthcare information systems, and life sciences solutions. The company's pharmacovigilance product line is centered on integrated safety solutions and regulatory compliance automation, and has a strong focus on data security and innovation to complement its strong market presence in both pharmaceutical and biotech environments.
-
In September 2024, introduced advanced pharmacovigilance analytics and machine learning-based adverse event prediction features for its Oracle Argus Safety platform, strengthening signal detection capabilities and expanding adoption among multinational pharmaceutical companies.
Pharmacovigilance Market Key Players:
-
IQVIA Holdings Inc.
-
Accenture plc
-
Oracle Corporation
-
PAREXEL International Corporation
-
Cognizant Technology Solutions
-
ArisGlobal LLC
-
Syneos Health
-
ICON plc
-
Pharmaceutical Product Development (PPD)
-
Laboratory Corporation of America (Labcorp Drug Development)
-
Wipro Limited
-
Capgemini SE
-
Medpace Holdings, Inc.
-
Freyr Solutions
-
Veeva Systems Inc.
-
Genpact Limited
-
BioClinica (Clario)
-
PRA Health Sciences
-
Pfizer CentreOne
-
Ergomed plc
-
TAKE Solutions Limited
-
Celegence (ITClinical)
-
United BioSource Corporation (UBC)
-
DrugSafety Services
-
SIRO Clinpharm
| Report Attributes | Details |
| Market Size in 2025 | US$ 8.91 Bn |
| Market Size by 2035 | US$ 24.69 Bn |
| CAGR | CAGR of 10.34% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By product Life Cycle • By Service Provider • By Type • By Process Flow • By Therapeutic • By End Use |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Eastern Europe [Poland, Romania, Hungary, Turkey, Rest of Eastern Europe] Western Europe] Germany, France, UK, Italy, Spain, Netherlands, Switzerland, Austria, Rest of Western Europe]). Asia Pacific (China, India, Japan, South Korea, Vietnam, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (Middle East [UAE, Egypt, Saudi Arabia, Qatar, Rest of Middle East], Africa [Nigeria, South Africa, Rest of Africa], Latin America (Brazil, Argentina, Colombia Rest of Latin America) |
| Company Profiles |
IQVIA Holdings Inc., Accenture plc, Oracle Corporation, PAREXEL International Corporation, Cognizant Technology Solutions, ArisGlobal LLC, Syneos Health, ICON plc, Pharmaceutical Product Development (PPD), Laboratory Corporation of America (Labcorp Drug Development), Wipro Limited, Capgemini SE, Medpace Holdings, Inc., Freyr Solutions, Veeva Systems Inc., Genpact Limited, BioClinica (Clario), PRA Health Sciences, Pfizer CentreOne, Ergomed plc, TAKE Solutions Limited, Celegence (ITClinical), United BioSource Corporation (UBC), DrugSafety Services, SIRO Clinpharm. |
Frequently Asked Questions
Ans: North America dominated the Pharmacovigilance Market in 2025.
Ans: By service provider, the Contract Outsourcing segment dominated the Pharmacovigilance Market in 2025.
Ans: Stringent Regulatory Requirements and Compliance Mandates are Driving the Pharmacovigilance Market Growth.
Ans: The Pharmacovigilance Market size was USD 8.91 billion in 2025 and is expected to reach USD 24.69 billion by 2035.
Ans: The Pharmacovigilance Market is expected to grow at a CAGR of 10.34% over the forecast period.

