Point-of-Care Molecular Diagnostics Market Report Scope & Overview:
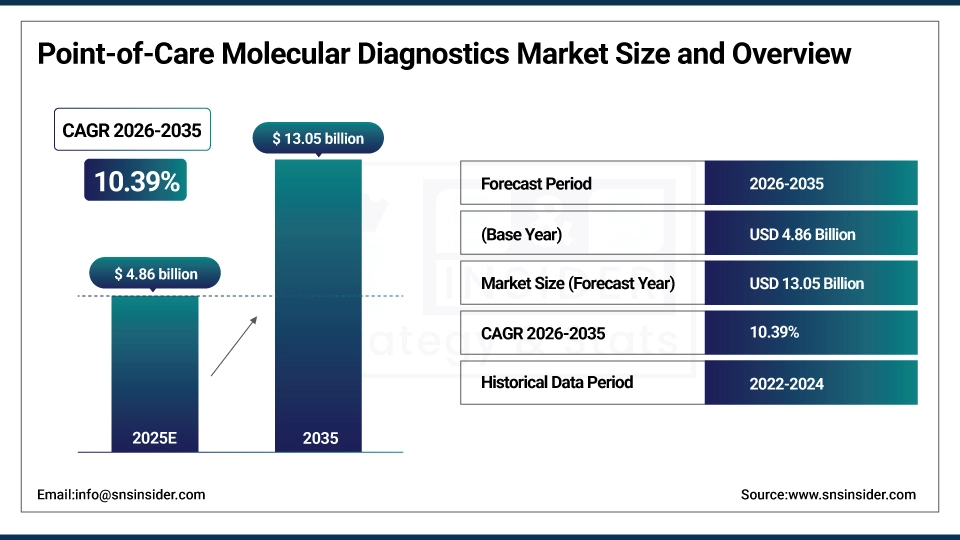
The Point-of-Care Molecular Diagnostics Market was valued at USD 4.86 Billion in 2025 and is expected to reach USD 13.05 Billion by 2035, growing at a CAGR 10.39% of from 2026-2035.
The rise in need for fast near-patient testing and early disease diagnosis in hospitals and clinics, as well as within the home, is considerably speeding up the Point-of-Care Molecular Diagnostics market. The increasing incidence of infectious and chronic diseases and decentralized healthcare services are factors driving the uptake of point-of-care (POC) molecular diagnostics systems. These advanced technologies are PCR-based tests, isothermal amplification, and lab-on-a-chip platforms which are utilized to provide higher accuracy tests, timely tests, and easier to use tests to improve clinical decision making by the healthcare profession and in turn improve patient outcomes.
The Point-of-Care Molecular Diagnostics market is growing at a CAGR of 9–10%, driven by demand for rapid near-patient testing, rising infectious and chronic diseases, and adoption of PCR-based, isothermal, and lab-on-a-chip technologies improving clinical outcomes.
Point-of-Care Molecular Diagnostics Market Size and Forecast
-
Market Size in 2025: USD 4.86 Billion
-
Market Size by 2035: USD 13.05 Billion
-
CAGR: 10.39%
-
Base Year: 2025
-
Forecast Period: 2026-2035
-
Historical Data: 2022-2024
To Get more information on Point-of-Care Molecular Diagnostics Market - Request Free Sample Report
Point-of-Care Molecular Diagnostics Market Trends
-
Rising demand for rapid near-patient testing: POC molecular diagnostics are increasingly adopted in hospitals, clinics, and home settings for faster disease detection.
-
Advancements in molecular technologies: PCR-based, isothermal amplification, and lab-on-a-chip platforms improve test accuracy, speed, and usability.
-
Decentralization of healthcare services: Growth in outpatient, ambulatory, and home testing drives broader adoption of POC systems.
-
Integration with digital health: Connectivity with healthcare IT and telemedicine platforms enables real-time data sharing and remote patient monitoring.
-
Expansion into infectious disease and chronic condition screening: Broadening applications increase market scope and clinical impact.
-
Increasing investment and commercialization: Accelerated development, regulatory approvals, and partnerships are driving market growth globally.
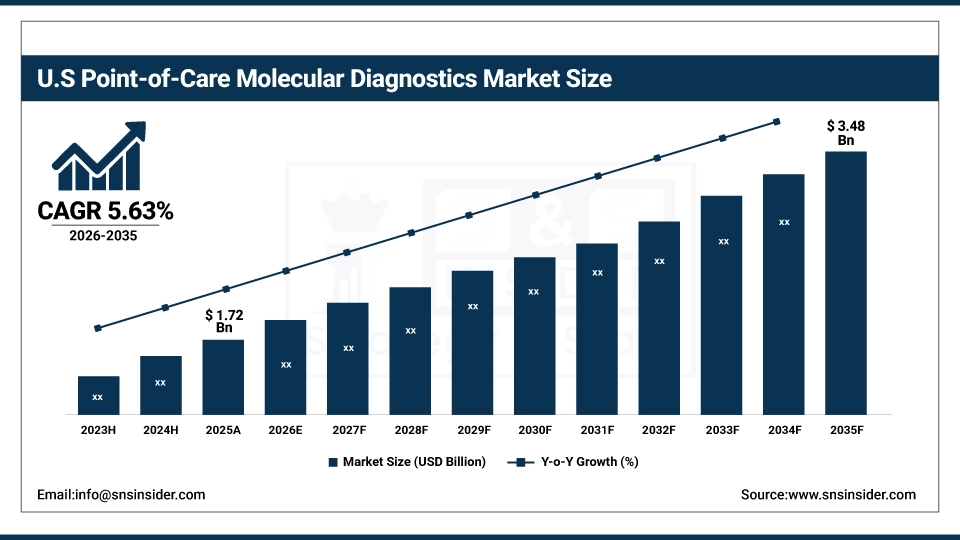
The U.S. Point-of-Care Molecular Diagnostics Market is projected to grow from USD 1.72 Billion in 2025 to USD 3.48 Billion by 2035, at a CAGR of 5.63%. Growth is driven by rising demand for rapid near-patient testing, increasing prevalence of infectious and chronic diseases, expansion of home and outpatient testing, continuous advancements in PCR, isothermal amplification, and lab-on-a-chip technologies, and increasing R&D and commercialization of POC molecular diagnostic solutions.
Point-of-Care Molecular Diagnostics Market Growth Drivers:
-
Rising Demand for Rapid Near-Patient Testing and Early Disease Detection
The demand for rapid and accurate testing in hospitals, clinics, and home setting drives the growth of the Point-of-Care Molecular Diagnostics market. Technical attributes like high test accuracy, fast response time, portability, and simplicity of use are emerging as major adoption attributes. According to research, by 2025, most healthcare providers and home-based testing solutions will incorporate PCR, isothermal amplification, and lab-on-chip platforms to improve clinical decision making, decrease the time to diagnosis, and improve patient outcome.
Point-of-Care Molecular Diagnostics Market Restraints:
-
High Development Costs and Regulatory Challenges Limiting Widespread Adoption
Limited high R&D and manufacturing costs for advanced POC molecular diagnostic systems (like cartridge-based kits, portable analyzers, and microfluidic platforms) are restraining the growth of the market. Complicated regulatory clearances, issues with standardization, and varied performance in different clinical environments continues to remain roadblocks as well. In addition, other diagnostic modalities, including centralized laboratory-based testing and rapid antigen assays, present a competitive alternative and have hampered the widespread adoption of molecular POC devices.
Point-of-Care Molecular Diagnostics Market Opportunities:
-
Expansion in Home Testing, Outpatient Clinics, and Emerging Markets
Market growth will mainly be driven by the increasing adoption of POC molecular diagnostics in home care settings, ambulatory centers and developing and resource-limited regions. The growing technological sophistication of miniaturized devices, multiplex tests, and integration with digital health platforms should continue increasing market penetration. Developments in lower cost, mobile, and highly accurate point-of-care molecular systems are anticipated to accelerate worldwide revenue growth and adoption through 2035.
Point-of-Care Molecular Diagnostics Market Segment Analysis:
-
By Technology: In 2025, PCR-based tests dominated with 50% share; isothermal amplification-based tests are projected to be the fastest-growing segment during 2026–2035.
-
By Application: In 2025, infectious diseases testing dominated with 45% share; oncology testing is projected to be the fastest-growing segment during 2026–2035.
-
By End Use: In 2025, hospitals held 55% share; home testing is expected to be the fastest-growing segment during 2026–2035.
-
By Component Type: In 2025, cartridge-based disposable kits led with 48% share; lab-on-a-chip platforms are projected to be the fastest-growing segment during 2026–2035.
By Technology: PCR-Based Tests Lead as Isothermal Amplification-Based Tests Emerge as Fastest-Growing Segment
The PCR-based tests segment dominated the technology segment due to high accuracy, reproducibility and widespread adoption in hospitals, clinics, and laboratory environments. Such tests are the gold standard to identify infectious pathogens and genetic markers with high reproducibility and speed. They enjoy market supremacy due to their robust nature, regulations approval, and compatibility with almost all clinical applications.
The largest growth in tests is being seen for isothermal amplification-based tests, due to the growing need for rapid, portable and inexpensive point-of-care molecular diagnostics. Low-cost, lab- and instrument-free procedures that have shorter turnaround times and are easier to use are appropriate for home testing, remote clinics, and resource-limited settings, making them well-suited to high-throughput decentralized tests to scale health care solution space.
By Application: Infectious Diseases Lead as Oncology Emerges as Fastest-Growing Segment
The largest application segment of this technology is infectious diseases testing—high-speed examination of infectious pathogens such as COVID-19, influenza, HIV, and TB is crucial for timely clinical decisions and outbreak management. It accounts for dominant share, due to high adoption in hospitals, emergency care and public health programs.
The Oncology testing segment will grow at the fastest CAGR during the forecast period, and high-volume demand for early cancer detection along with growing interest in genetic biomarkers screening and personalized medicine are boosting segment growth. Fast adoption of POC molecular diagnostics in outpatient clinics, home based monitoring and targeted therapy selection drives market growth in this segment.
By End Use: Hospitals Lead as Home Testing Emerges as Fastest-Growing Segment
In the end-use segment, hospitals represent the largest share: hospitals are the main centers for clinical diagnostics providing adequately trained personnel, standard workflows, and infrastructure that assures regulatory compliance. They have a high patient volume, testing protocols can replicate quickly, and they can integrate into existing hospital IT systems.
Among the end-use segments, home testing is anticipated to be the fastest growing segment due to increasing preference for rapid and self testing of molecular tests by patients. Point-of-Care Advances in simple kits, linkages to digital health platforms, and the broadening of telemedicine are hastening adoption in home and ambulatory settings
By Component Type: Cartridge-Based Disposable Kits Lead as Lab-on-a-Chip Platforms Emerge as Fastest-Growing Segment
The cartridge-based disposable kit segment, accounting for the largest share of the segment type, is highly preferred due to ease of use, similar workflows, and consistent sample handling. The design is convenient with minimal potential contamination, high sample throughput, and is suitable to be integrated with automated analyzers for hospital and clinic applications.
The fastest growing segment is lab-on-a-chip platforms, the ability to mix multiple tests in one place, with rapid turnaround and portable nature. This is leading to increasing adoption of these platforms in home testing, mobile clinics, and point-of-need settings, enabled by developments in microfluidics, automated sample handling, and integration with digital health systems.
Regional Analysis
North America Point-of-Care Molecular Diagnostics Market Insights:
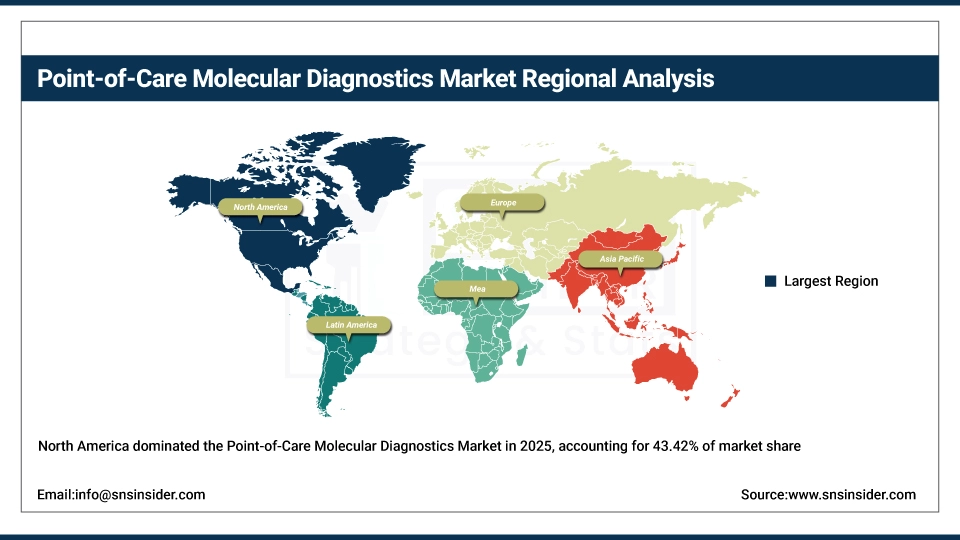
The shares of the North America market reached 43.42% in 2025, ranking the first in the global market. This is attributed to the presence of advanced healthcare infrastructure, high adoption of rapid diagnostic testing, and strong presence of leading POC molecular diagnostics companies in the region. The U.S. and Canada are top contenders for tech development, regulatory approvals, and early adoption of advanced POC systems. The ability to deploy widely in hospitals and clinics and beyond, as a tool for home health and telemedicine platforms, undergirds that market share leadership from 2026 through to 2035.
Get Customized Report as per Your Business Requirement - Enquiry Now
Asia-Pacific Point-of-Care Molecular Diagnostics Market Insights:
Asia-Pacific occupies a rapidly growing site in the Point-of-Care Molecular Diagnostics market, and is expected to grow with a CAGR of 15.82% during 2026-2035. The region is projected to witness the fastest growth due to the increasing usage of home testing, outpatient clinics, and decentralized healthcare services. Market Introduction Technological advancements in PCR, isothermal amplification and lab-on-a-chip platforms, increasing healthcare expenditure along with increasing awareness regarding early disease detection are some of the few key factors driving the growth of this market. Emerging POC molecular diagnostics adoption and commercialization activity in China, India, Japan, and Southeast Asian countries will drive regional growth and contribute to overall market expansion.
Europe Point-of-Care Molecular Diagnostics Market Insights:
Europe has been able to drive market growth by enforcing strict regulatory standards, high healthcare spending, and increased adoption of decentralized diagnostics. Hospital networks, better laboratories and increased adoption of POC molecular diagnostics for infectious disease and oncology testing all encourage adoption. Both Western and Eastern Europe display consistent growth, powered by solid R&D performances and modernization of healthcare systems.
Latin America Point-of-Care Molecular Diagnostics Market Insights:
The Latin America market is growing at a steady pace, primarily on account of growing healthcare awareness, rising prevalence of infectious diseases, and gradual adoption of point-of-care (POC) molecular diagnostic technologies in hospitals and clinics. Expansion of governmental programs, rising investment toward outpatient services, and easy accessibility of rapid testing platforms are ascribed to support the growth of market in Brazil, Mexico and Argentina.
Middle East & Africa Point-of-Care Molecular Diagnostics Market Insights:
Promising growth is observed in the Middle East & Africa market due to the demand for rapid diagnostics and increasing healthcare investment and gradually improving laboratory infrastructure in the region. The rapid adoption of EMR software in hospitals, clinics and home healthcare is the primary driver behind its expansion, with the United Arab Emirates, Saudi Arabia and South Africa, among others, playing critical roles. Meanwhile, global market prospects of the regional market is also benefitting from the balancing regulatory frameworks and investment in digital healthcare
Point-of-Care Molecular Diagnostics Market Competitive Landscape:
Abbott Laboratories, based in Abbott Park, Illinois, USA, is a leading global healthcare company with a strong presence in the Point‑of‑Care Molecular Diagnostics market; it specializes in rapid diagnostic platforms such as the ID NOW system, which delivers fast PCR or isothermal amplification‑based molecular test results for infectious diseases and other conditions.
-
In 2025, Abbott announced its planned acquisition of Exact Sciences for up to USD 23 billion, marking a major expansion into cancer screening and broader molecular diagnostics capabilities, strengthening its diagnostic portfolio and long‑term growth potential.
Roche Diagnostics, headquartered in Basel, Switzerland, is a major global developer of diagnostic systems including its cobas Liat and cobas Pulse POC molecular platforms, which offer highly accurate PCR‑grade testing for infectious diseases and other clinical applications.
-
In 2025, Roche received FDA 510(k) clearance and CLIA waiver for its cobas Liat STI multiplex assay panels, enabling rapid decentralized testing for sexually transmitted infections and expanding its POC molecular diagnostics offerings.
Danaher Corporation (Cepheid), based in Washington, D.C., USA, through its subsidiary Cepheid, is a pioneer in molecular diagnostics with the GeneXpert Xpress platform, which delivers rapid PCR‑based testing for respiratory infections, tuberculosis, and sexually transmitted diseases in decentralized clinical environments.
-
In June 2025, Cepheid announced that Health Canada issued a medical device licence for its Xpert HIV‑1 Viral Load XC test, broadening its test coverage for HIV viral load monitoring and enhancing its presence in global POC diagnostics.
Point-of-Care Molecular Diagnostics Market Market Key Players
-
Abbott Laboratories
-
Roche Diagnostics
-
Danaher Corporation (Cepheid)
-
bioMérieux SA
-
QIAGEN N.V.
-
Thermo Fisher Scientific
-
QuidelOrtho Corporation
-
Hologic
-
Becton Dickinson (BD)
-
Bio-Rad Laboratories
-
Meridian Bioscience
-
Biocartis Group NV
-
Co-Diagnostics
-
Binx Health
-
Molbio Diagnostics Pvt. Ltd.
-
GenMark Diagnostics
-
Siemens Healthineers
-
Orasure Technologies
-
Visby Medical
-
AccuBioTech Co. Ltd.
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 4.86 Billion |
| Market Size by 2035 | USD 13.05 Billion |
| CAGR | CAGR of 10.39% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Technology: (PCR-based tests, Isothermal amplification-based tests, Sequencing-based tests) • By Application: (Infectious diseases, Oncology, Antimicrobial resistance testing) •By End User: (Hospitals, Home testing, Ambulatory facilities) • By Component Type: (Cartridge-based Disposable kits, Portable instruments, Lab-on-a-chip platforms) |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
| Company Profiles | Abbott Laboratories, Roche Diagnostics, Danaher Corporation (Cepheid), bioMérieux SA, QIAGEN N.V., Thermo Fisher Scientific, QuidelOrtho Corporation, Hologic, Becton Dickinson (BD), Bio-Rad Laboratories, Meridian Bioscience, Biocartis Group NV, Co-Diagnostics, Binx Health, Molbio Diagnostics Pvt. Ltd., GenMark Diagnostics, Siemens Healthineers, Orasure Technologies, Visby Medical, AccuBioTech Co. Ltd. |

