Tuberculosis Therapeutics Market Report Scope & Overview:
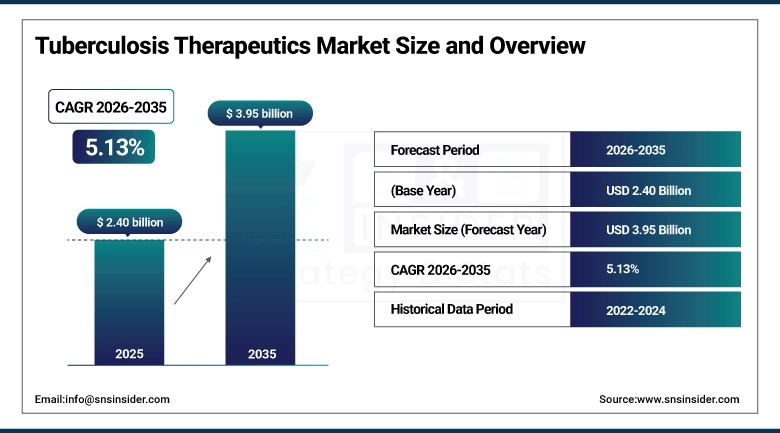
The Tuberculosis Therapeutics Market size was valued at USD 2.40 billion in 2025 and is expected to reach USD 3.95 billion by 2035, growing at a CAGR of 5.13% over the forecast period of 2026-2035.
The market for tuberculosis therapeutics is influenced by the critical and consistent need for tuberculosis pharmaceuticals. This is mainly due to the continuous prevalence of tuberculosis across the globe, the continuous rise of drug-resistant tuberculosis cases, and the rapid focus of governments and international agencies to implement tuberculosis elimination programs. According to the World Health Organization's Global Tuberculosis Report 2023, 10.6 million people contracted tuberculosis in 2022. Furthermore, tuberculosis claimed 1.3 million lives in 2022. It is evident that tuberculosis is one of the most prevalent infectious diseases causing death across the globe. The continuous prevalence of tuberculosis is causing a consistent pharmaceutical market for tuberculosis drugs, including first-line drugs such as isoniazid, rifampicin, ethambutol, pyrazinamide, second-line drugs such as bedaquiline, delamanid, linezolid, pretomanid, and others for drug-resistant tuberculosis cases. The market is also influenced by the World Health Organization's END TB Strategy to decrease tuberculosis incidence by 90 percent and tuberculosis deaths by 95 percent in 2030, as compared to 2015.
For instance, in 2023, the Global Fund committed USD 3.6 billion specifically to TB programs worldwide across its 2023-2025 replenishment cycle, directly stimulating demand for both innovator and generic anti-tuberculosis therapeutics in high-burden countries.
Tuberculosis Therapeutics Market Size and Forecast:
-
Market Size in 2025: USD 2.40 billion
-
Market Size by 2035: USD 3.95 billion
-
CAGR: 5.13% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get more information on Tuberculosis Therapeutics Market - Request Free Sample Report
Tuberculosis Therapeutics Market Trends
-
Accelerating adoption of shorter treatment regimens such as the 6-month BPaL (bedaquiline, pretomanid, linezolid) regimen for MDR-TB, reducing treatment duration from 18-24 months, improving patient adherence and therapeutic outcomes.
-
Growing integration of WHO-prequalified generic anti-TB drugs from emerging market manufacturers, lowering per-patient treatment costs and improving drug accessibility in low- and middle-income countries.
-
Rising demand for fixed-dose combination (FDC) tablets, which simplify dosing schedules, reduce pill burden, and improve medication adherence rates among active TB patients in community health settings.
-
Increased investment in the TB drug pipeline, with more than 20 drug candidates in active clinical trials as of 2024, targeting novel mechanisms of action to address drug-resistant TB strains.
-
Expansion of child-appropriate dispersible tablet formulations and pediatric dosing options, supported by global health initiatives addressing the significant treatment gap in TB-affected children under 15 years of age.
-
Growing emphasis on latent TB infection (LTBI) treatment using shorter regimens such as 3HP (isoniazid plus rifapentine once weekly for 12 weeks), reducing progression to active TB disease in high-risk populations.
-
Strong government procurement programs, USAID-funded drug supply chains, and national TB program tenders driving bulk purchase volumes and market-level revenue growth across Asia, Africa, and Latin America.
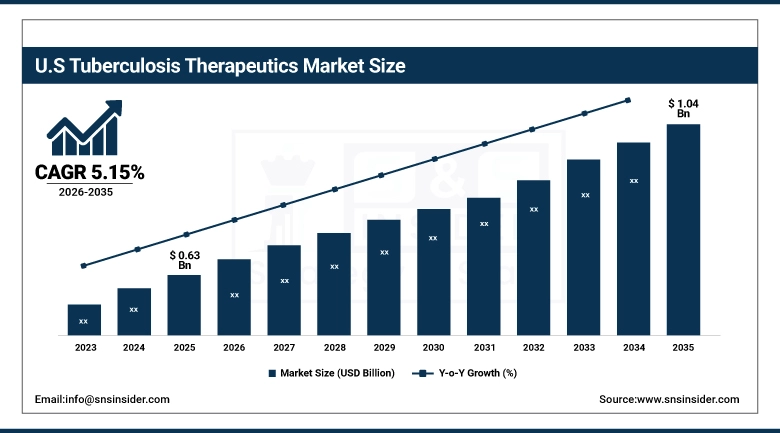
The U.S. Tuberculosis Therapeutics Market was valued at USD 0.63 billion in 2025 and is expected to reach USD 1.04 billion by 2035, growing at a CAGR of 5.15% from 2026-2035. The United States represents a substantial but relatively developed market for TB drugs, with low domestic TB incidence rates of approximately 9,000 cases reported by the US CDC as of 2023. This market has been sustained by robust public health funding, federally mandated completion programs, as well as substantial USAID-funded international procurement activity. The availability of new drug approvals under the FDA's accelerated approval process for MDR TB drugs, as well as the rising incidence rates of latent TB among immigrant populations at risk, will continue to fuel this market.
Tuberculosis Therapeutics Market Growth Drivers:
-
Rising MDR-TB and XDR-TB Incidence is Driving Tuberculosis Therapeutics Market Growth
The increasing global incidence of multidrug-resistant tuberculosis (MDR-TB) and extensively drug-resistant tuberculosis (XDR-TB) is a major growth driver in the tuberculosis therapeutics market. WHO states that in 2022, an estimated 410,000 new MDR/RR-TB cases were reported worldwide. However, only 2 out of 5 people with drug-resistant TB are receiving appropriate treatment. The clinical challenges in treating MDR-TB patients with 18 to 24 months of second-line drug regimens with severe side effects are fuelling the need for newer, shorter regimens with better efficacy. Regulatory approval for bedaquiline, pretomanid, and delamanid by FDA and EMA and WHO guideline recommendations have significantly increased the second-line drug market in the global tuberculosis therapeutics market share.
For instance, in November 2024, WHO updated its MDR-TB treatment guidelines to prioritize the 6-month BPaL regimen for eligible patients, directly boosting demand for bedaquiline-containing regimens across 45 high-burden countries participating in the WHO-coordinated procurement programs.
Tuberculosis Therapeutics Market Restraints:
-
Donor-Dependent Funding Volatility and Limited Domestic Financing are Hampering the Tuberculosis Therapeutics Market Growth
One of the most important structural limitations for the tuberculosis therapeutics market is the reliance of high-burden, low-income countries upon external donor funding from organizations such as the Global Fund to Fight AIDS, Tuberculosis, and Malaria; USAID; and bilateral aid programs to support national drug procurements for tuberculosis treatment. According to WHO estimates, for many of these high-burden countries, less than 50% of total tuberculosis program expenditure is funded domestically. This is a critical issue that causes instability in drug procurements, creates uncertain revenues for drug companies, and causes drug stock-outs that affect continuity of therapy. Political changes that affect donor country budgets, geopolitical events, and other emergencies that compete for donor dollars can limit TB treatment markets at certain times.
Tuberculosis Therapeutics Market Opportunities:
-
Novel Drug Approvals and Shortened Regimen Development Present Significant Growth Opportunities for the Tuberculosis Therapeutics Market
The tuberculosis therapeutics market has opportunities in new drug development and approval of new, shorter, and safer regimens of drug-sensitive as well as drug-resistant tuberculosis. The active clinical pipeline includes new drugs in development, e.g., sutezolid, TBA-7371, and OPC-167832, which target new bacterial targets. Regulatory incentives, e.g., FDA priority review vouchers for neglected tropical disease drugs, EMA accelerated assessment, and WHO prequalification, are also providing encouragement for investment in the market by biopharmaceutical companies. Furthermore, the vast market potential in the treatment of latent tuberculosis, which has been estimated at more than 1.7 billion individuals, provides a new opportunity in the prevention of tuberculosis using new, short regimens as advocated by WHO guidelines.
For instance, in March 2024, the TB Alliance reported that its DELIBERATE Phase 3 trial results confirmed the efficacy of the BPaL regimen in reducing treatment duration for XDR-TB to 6 months with a treatment success rate of 89%, representing a breakthrough that is expected to accelerate regulatory submissions across multiple jurisdictions in 2025 and 2026.
Tuberculosis Therapeutics Market Segment Analysis
-
By disease type, active TB held the largest share of approximately 68.45% in 2025, while the latent TB segment is expected to register the highest growth with a CAGR of 6.17%.
-
By therapeutics therapy, first-line therapy dominated the market with around 65.72% share in 2025, while the second-line therapy segment is expected to register the highest growth with a CAGR of 6.89%.
-
By route of administration, oral accounted for the leading share of approximately 72.34% in 2025, and is expected to register the highest CAGR of 5.47%.
-
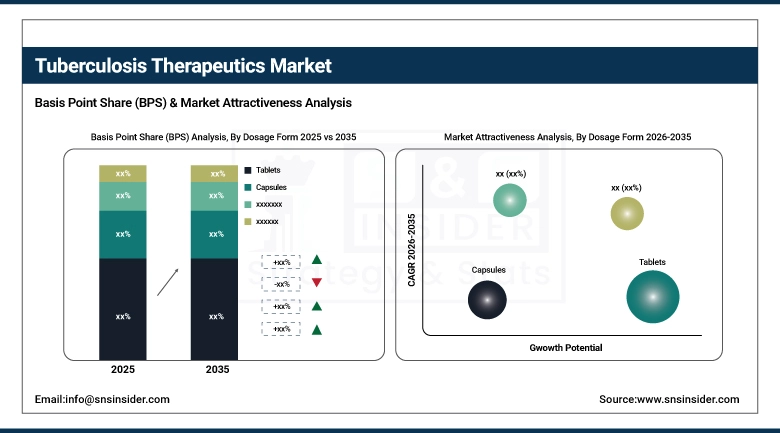
By dosage form, tablets held the largest share of around 56.28% in 2025, while the injection segment is expected to grow at a notable CAGR of 5.82%.
-
By distribution channel, hospital pharmacy led with approximately 61.47% share in 2025, and online pharmacy is expected to register the highest growth with a CAGR of 7.24%.
By Dosage Form, Tablets Lead, While Injections Register Notable Growth
Tablets dominate segmental share and accounted for 56.28% market share in 2025, as these are the preferred dosage forms of all the categories for TB treatment. This counts with the FDCs because you know FDCs are easy to use, stable in formulation and ideal for bulk purchases by the government especially in resource limited countries. The injection segment is projected to grow at a significant CAGR of 5.82% due to the continued use of injectables for MDR TB patients who remain hospitalized in high-burden countries where parenteral options remain part of treatment protocols until full guideline adoption.
By Disease Type, Active TB Leads the Market, While Latent TB Registers Fastest Growth
By 2025, the highest share in the market came from the active TB segment with a share of nearly 68.45%. This reflects the immediate pharmacotherapeutic need, the treatment regimens standardized by national TB programs, and the high volume of drug procurement for notified active TB cases. Health agencies have made effort at active case management with regular procurements of drugs, ascertained by government regulations and WHO DOTS strategies. However, the latent TB segment is anticipated to be the fastest-growing segment with a CAGR of 6.17% during the forecast period (2026-2035). This is because of the intensification of global activities on preventive therapy, broader LTBI screening among high-risk groups such as health care workers, HIV-infected individuals, and immunocompromised patients and WHO recommendations for 3HP and 1HP regimens for short-course treatment for maximal treatment completion.
By Therapeutics Therapy, First-Line Therapy Dominates, While Second-Line Therapy Shows Rapid Growth
In 2025, first-line therapy held around 65.72% share of market revenue due the global burden of drug-susceptible TB cases along with global acceptance of isoniazid, rifampicin, ethambutol and pyrazinamide-based regimens backed by national TB program and international procurement. The second-line therapy segment is expected to grow at the largest CAGR of 6.89% during 2026-2035 due to the anticipated rise in the number of diagnosed MDR-TB and XDR-TB cases over the forecast years, the recent WHO guidelines recommending all-oral second-line regimens, and the growing programmatic uptake of newer drugs (such as bedaquiline, linezolid, clofazimine, cycloserine, and delamanid) globally.
By Route of Administration, Oral Leads and Registers Highest Growth
Oral route of administration led the pack with a total of 72.34% tuberculosis therapeutics market share in 2025, which is in accordance with WHO recommendations on the use of all oral regimens for first-line and second-line tuberculosis treatment. Parenteral route, which is used in the treatment of severe and disseminated forms of tuberculosis, and injectable forms of second-line antitubercular drugs like amikacin and streptomycin, hold the secondary position. The trend towards non-injectable aminoglycosides after the WHO guideline changes in 2022 has strengthened the oral segment, which is expected to hold the highest CAGR of 5.47% in the tuberculosis therapeutics market by 2035.
By Distribution Channel, Hospital Pharmacy Leads, While Online Pharmacy Registers Fastest Growth
Hospital pharmacy, as the distribution channel, held a share of roughly 61.47% in distribution channel in 2025, which is in line with the clinical complexity of TB management as both drug-susceptible and drug-resistant cases require initiation of treatment in hospital, directly observed therapy and adverse effect monitoring. The online pharmacy channel has a growth potential with the highest CAGR of 7.24% during the period of 2026-2035; growth is likely to be powered by expansion of e-pharmacy platforms in India, Brazil and other countries in South-East Asia, digital health integration with national TB notification systems and availability of latent TB preventive therapy through outpatient and community health dispensing models.
Tuberculosis Therapeutics Market Regional Highlights:
Asia Pacific Tuberculosis Therapeutics Market Insights:
Asia Pacific is the fastest-growing regional segment in the tuberculosis therapeutics market. This market is expected to register a CAGR of 6.42% during the forecast period. This region has the largest share in the global tuberculosis cases. India contributes about 27% of all global tuberculosis cases. Indonesia, China, the Philippines, and Pakistan come next in the list. These five countries account for more than 57% of the new cases of tuberculosis reported worldwide. Asia Pacific is both the largest and fastest-growing tuberculosis market. India's National TB Elimination Programme aims to eliminate tuberculosis by 2025, which is earlier than the global target of 2030. China also has action plans to control tuberculosis. Indonesia and the Philippines also have high levels of funding from the global fund. All these factors are boosting the tuberculosis market in the Asia Pacific region in terms of both quantity and pharmaceutical production.
North America Tuberculosis Therapeutics Market Insights:
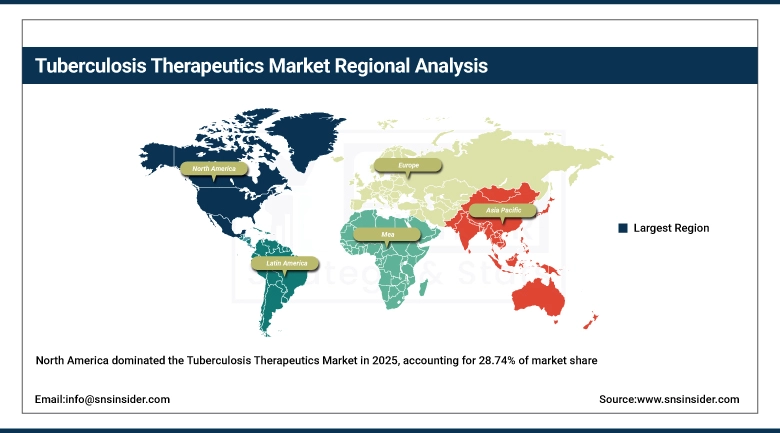
In 2025, North America held the largest share at 28.74% due to high innovator drug prices in the region, high levels of international drug procurements funded by USAID by organizations located in the US, and high levels of domestic R&D investment in new drug development for TB in the region. Even with its high burden of TB cases, this region will continue to be the highest revenue-generating region, sustained by having its major innovator drug manufacturers (Johnson & Johnson and Pfizer), FDA than grants accelerated approval pathways for TB drugs, and very large NIH R&D investments.
Get Customized Report as per Your Business Requirement - Enquiry Now
Europe Tuberculosis Therapeutics Market Insights:
Europe accounts for the second-largest TB drugs market as it suffers from a high incidence rate, especially in Eastern Europe where WHO European Region reportedly faced some of the highest MDR-TB rates worldwide with Russia, Ukraine and Moldova having contributed disproportionately to global drug-resistant TB cases. Europe remains ahead of the market as EMA gives bedaquiline and delamanid approvals as robust health ministry budgets for procurement in Germany, France, and the UK as well as strong pharmacovigilance systems for the safe use of second-line drugs.
Latin America (LATAM) and Middle East & Africa (MEA) Tuberculosis Therapeutics Market Insights:
Latin America and Middle East & Africa tuberculosis therapeutics market is backed up with the support for TB control programs, donor funded drug procurement and growing domestic pharmaceutical manufacturing capabilities in the region. The main market driver in the region is Brazil - with an estimated 96,000 new cases notified in 2022, representing 25% of all new TB cases in the 30 high TB burden countries in the world, Brazil is the country with most TB burden in Latin America. In the MEA region South Africa has the highest TB incidence rate per capita globally, at 513 cases per 100,000 population, and while only around one-third of TB cases are attributable to HIV co-infection, the high rates of HIV in the country creates the continued need for TB-HIV co-treatment regimens, making South Africa a key market for both first line and second line anti-tuberculosis therapeutics.
Tuberculosis Therapeutics Market Competitive Landscape:
Johnson & Johnson (Janssen Pharmaceuticals) is the innovator behind bedaquiline (Sirturo), the first novel TB drug approved in over 40 years, which remains a cornerstone of WHO-recommended MDR-TB treatment regimens globally. The company continues to invest in access programs, voluntary licensing agreements, and regulatory submissions to expand bedaquiline availability in high-burden countries.
-
In January 2025, Janssen expanded its voluntary licensing agreement for bedaquiline to 44 countries through the Medicines Patent Pool, enabling production of generic bedaquiline by multiple manufacturers and improving affordability and supply security for national TB programs in high-burden low-income countries.
Otsuka Pharmaceutical Co., Ltd. is the developer of delamanid (Deltyba), a nitroimidazole approved for MDR-TB treatment in adults, adolescents, and children, and continues to conduct post-marketing commitments and pediatric indication expansions globally.
-
In May 2024, Otsuka Pharmaceutical received WHO prequalification for its 25 mg and 50 mg delamanid tablet formulations, enabling broader procurement through UNITAID and Global Fund-financed supply chains across Sub-Saharan Africa and Southeast Asia.
Lupin Limited is one of the world's largest manufacturers of anti-tuberculosis drugs, producing a comprehensive portfolio of first-line and fixed-dose combination (FDC) TB products that are WHO-prequalified and supplied to over 30 countries through government and international procurement channels.
-
In August 2024, Lupin Limited received regulatory approval from India's CDSCO for an optimized fixed-dose combination tablet formulation for pediatric TB treatment, aligning with WHO's child-friendly medicine guidelines and targeting the significant pediatric TB treatment gap in South Asia and Sub-Saharan Africa.
Tuberculosis Therapeutics Market Key Players:
-
Johnson & Johnson (Janssen Pharmaceuticals)
-
Otsuka Pharmaceutical Co., Ltd.
-
Lupin Limited
-
Pfizer Inc.
-
Novartis AG
-
Sanofi S.A.
-
Viatris Inc.
-
Cipla Limited
-
Sun Pharmaceutical Industries Ltd.
-
Aurobindo Pharma Ltd.
-
Macleods Pharmaceuticals Ltd.
-
Hikma Pharmaceuticals PLC
-
Laurus Labs Ltd.
-
Mankind Pharma Ltd.
-
GlaxoSmithKline PLC
-
Daiichi Sankyo Co., Ltd.
-
Strides Pharma Science Ltd.
-
Alembic Pharmaceuticals Ltd.
-
Akums Drugs & Pharmaceuticals Ltd.
-
Astellas Pharma Inc.
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 2.40 Billion |
| Market Size by 2035 | USD 3.95 Billion |
| CAGR | CAGR of 5.13% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Disease Type (Active TB, Latent TB) • By Therapeutics Therapy (First-Line Therapy, Second-Line Therapy) • By Route of Administration (Oral, Parenteral, and Others) • By Dosage Form (Tablets, Capsules, Injection, and Others) • By Distribution Channel (Hospital Pharmacy, Retail Pharmacy, and Online Pharmacy) |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, Poland, Turkey, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Rest of Latin America) |
| Company Profiles | Johnson & Johnson (Janssen Pharmaceuticals), Otsuka Pharmaceutical Co., Ltd., Lupin Limited, Pfizer Inc., Novartis AG, Sanofi S.A., Viatris Inc., Cipla Limited, Sun Pharmaceutical Industries Ltd., Aurobindo Pharma Ltd., Macleods Pharmaceuticals Ltd., Hikma Pharmaceuticals PLC, Laurus Labs Ltd., Mankind Pharma Ltd., GlaxoSmithKline PLC, Daiichi Sankyo Co., Ltd., Strides Pharma Science Ltd., Alembic Pharmaceuticals Ltd., Akums Drugs & Pharmaceuticals Ltd., Astellas Pharma Inc. |
Frequently Asked Questions
The Tuberculosis Therapeutics Market growth is driven by rising MDR-TB cases (410,000 cases in 2022), increasing adoption of shorter regimens like BPaL, and strong global funding such as USD 3.6 billion from the Global Fund (2023–2025).
The Tuberculosis Therapeutics Market was valued at USD 2.40 billion in 2025 and is projected to reach USD 3.95 billion by 2035, reflecting steady industry expansion.
The Tuberculosis Therapeutics Market is expected to grow at a CAGR of 5.13% from 2026 to 2035, driven by increasing TB incidence and drug innovation.
In the Tuberculosis Therapeutics Market, the active TB segment dominated with a 68.45% market share in 2025, due to high treatment demand and global TB burden.
The Tuberculosis Therapeutics Market is fastest growing in Asia Pacific, with a CAGR of 6.42%, supported by high TB prevalence and strong government initiatives.

