Virus Filtration Market Report Scope & Overview:
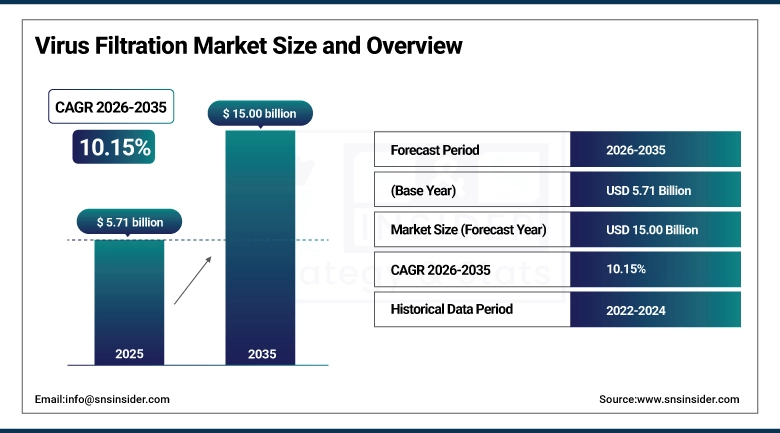
The Virus Filtration Market size was valued at USD 5.71 billion in 2025 and is expected to reach USD 15.00 billion by 2035, growing at a CAGR of 10.15% over the forecast period of 2026-2035.
Increasing demand from biopharmaceutical and biotechnology industries for comprehensive virus clearance technology due to the need for product safety of biologics, vaccines, blood products, and advanced therapy medicinal products, is bolstering global virus filtration market growth. This, along with greater attention of regulatory authorities like FDA, EMA, and ICH on virus safety validation, along with higher adoption rate for single-use bioprocessing technology and continuous processing are responsible for strengthening the market for virus retentive filtration technology and chromatography-based virus clearance technology. However, the need for minimal acquisition of viral-free products is driving the demand for virus filtration technology which includes, the growing approval rate of biologic drugs, increasing funding for cellular and gene therapy products, and an increasing trend toward outsource as well as contract manufacturing and research in this field is anticipated to further upsurge the market space for virus filtration technology even more.
For instance, in 2024, the FDA approved a record 73 novel biologics and biosimilars, generating a direct increase in demand for viral clearance validation services and virus-retentive filtration consumables across U.S.-based contract development and manufacturing organizations (CDMOs).
Virus Filtration Market Size and Forecast:
-
Market Size in 2025: USD 5.71 billion
-
Market Size by 2035: USD 15.00 billion
-
CAGR: 10.15% from 2026 to 2035
-
Base Year: 2025
-
Forecast Period: 2026–2035
-
Historical Data: 2022–2024
To Get more information on Virus Filtration Market - Request Free Sample Report
Virus Filtration Market Trends
-
Surging adoption of single-use virus filtration assemblies in biopharmaceutical manufacturing to minimize cross-contamination risks, reduce cleaning validation burdens, and accelerate production timelines.
-
Integration of virus-retentive nanofiltration membranes with downstream bioprocessing workflows, supporting higher throughput viral clearance with consistent log reduction values (LRVs) above 4.0 log10 for parvoviruses.
-
Expansion of continuous bioprocessing models driving demand for inline viral filtration modules that support real-time process monitoring and automated flow control.
-
Increasing application of virus filtration in cell and gene therapy manufacturing, including lentiviral and adeno-associated virus (AAV) vector production, fueling specialized hollow-fiber filtration technology adoption.
-
Growth of modular and prefabricated bioprocessing facilities in emerging markets, creating new opportunities for standardized virus filtration skids and plug-and-play filtration systems.
-
Rising regulatory emphasis on adventitious agent testing and viral clearance studies for biologics, accelerating uptake of GMP-grade filtration consumables and third-party validation services.
-
Growing use of artificial intelligence and process analytical technology (PAT) tools for predictive filter fouling detection and real-time optimization of viral filtration parameters.
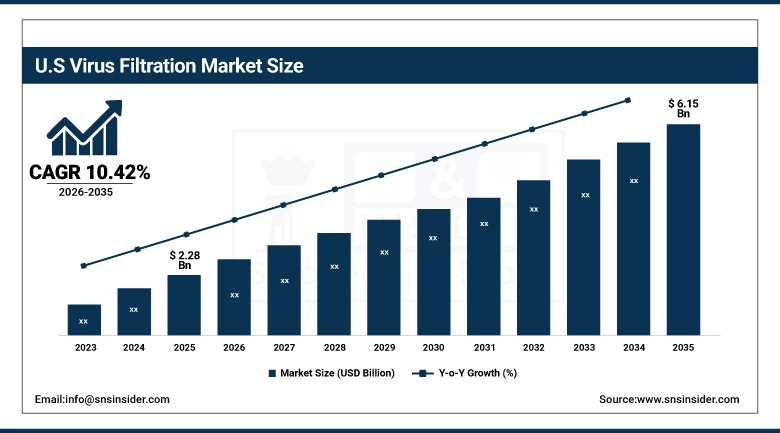
The U.S. Virus Filtration Market was valued at USD 2.28 billion in 2025 and is expected to reach USD 6.15 billion by 2035, growing at a CAGR of 10.42% from 2026 to 2035. The United States accounts for the highest percentage of the global virus filtration market, which can be largely attributed to the highly developed biopharmaceutical industry, FDA regulations related to viral safety, and the significant investment in the research and development of biologics and cell and gene therapies. The preeminent presence of CDMOs, research institutions, and biotech companies related to biopharma clusters in the United States, combined with significant funding by the NIH for research related to infectious diseases and vaccines, is resulting in the growth of highly advanced virus filtration technology.
Virus Filtration Market Growth Drivers:
-
Rising Biologics Production and Viral Safety Regulatory Requirements are Driving the Virus Filtration Market Growth
The rising global market for monoclonal antibodies, recombinant proteins, vaccines, and plasma-derived products is one of the main drivers for the virus filtration market. The regulatory bodies such as the FDA, EMA, and WHO have emphasized the need for robust viral clearance studies to be conducted as a prerequisite for approval of biological products. This has created a consistent market for virus-retentative filters, validation test kits, and downstream chromatography products. The development of biosimilars is another factor that has increased the market for virus filtration products. The development of COVID-19 vaccines using mRNA technology is another factor that will contribute to the growth of the virus filtration market.
For instance, in March 2024, global biologic drug revenues surpassed USD 420 billion, with virus filtration consumables accounting for approximately 18% of total downstream bioprocessing supply expenditures among top-20 pharmaceutical manufacturers.
Virus Filtration Market Restraints:
-
High Cost of Virus Filtration Validation and Limited Skilled Workforce are Hampering the Virus Filtration Market Growth
The high capital costs associated with virus filtration validation studies, especially those related to model viruses, as well as biosafety level 2 laboratory setup and regulatory affairs, can be a challenge, especially for small- and mid-sized biopharmaceutical companies as well as academic research centers. The high cost of a single viral clearance validation study can range from USD 150,000 to USD 400,000. Furthermore, the lack of expertise among bioprocess engineers in the field of viral safety evaluation and GMP filtration technology can be a challenge, especially in emerging countries, as the biopharmaceutical industry is in its developmental stages.
Virus Filtration Market Opportunities:
-
Cell and Gene Therapy Manufacturing Expansion Creates High-Value Growth Opportunities for the Virus Filtration Market
The rapid growth in cell and gene therapy pipelines offers a revolutionary opportunity to virus filtration technology suppliers. The production processes of AAV and LV viral vectors require specific tangential flow filtration and depth filtration products to guarantee the concentration, purity, and clearance of adventitious agents in viral vectors. As the world has over 3,500 active gene therapy trials in place, with over 40 gene therapy products approved or under rolling review by major regulatory bodies, the need to accelerate virus filtration is growing. Investment in specific viral vector CDMOs and the expansion of academic spin-out facilities have created a growing commercial need for virus filtration products and platforms.
For instance, in January 2024, the Alliance for Regenerative Medicine reported that global cell and gene therapy investment reached USD 9.8 billion in 2023, with viral vector manufacturing infrastructure accounting for over 31% of total facility capital expenditures, directly expanding the addressable market for virus filtration products.
Virus Filtration Market Segment Analysis
-
By product, consumables held the largest share of approximately 57.42% in 2025, while the services segment is expected to register the highest growth with a CAGR of 11.23%.
-
By technology, the filtration segment dominated the market with around 61.85% share in 2025, while the chromatography segment is expected to register the highest growth with a CAGR of 11.47%.
-
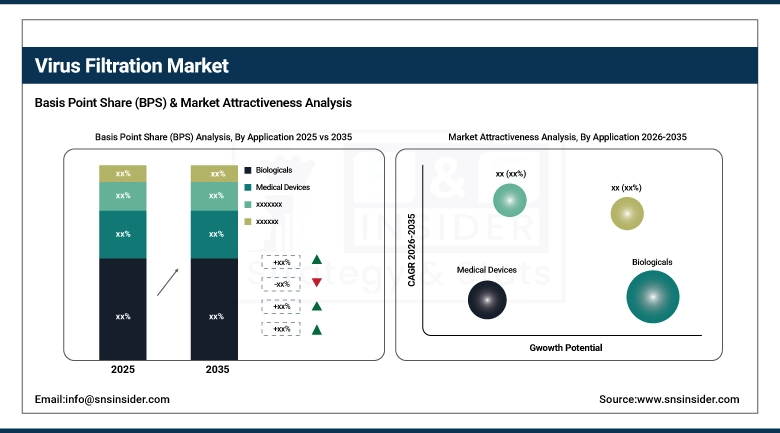
By application, the biologicals segment accounted for the leading share of nearly 64.32% in 2025 and is expected to register the highest growth with a CAGR of 11.06%.
-
By end-use, biopharmaceutical and biotechnology companies held the dominant share of approximately 54.71% in 2025, while contract research organizations are expected to register the highest CAGR of 11.84%.
By Application, Biologicals Lead and Register Fastest Growth
The largest share of 64.32% in 2025 was attributed to the biologicals application segment due to strong demand in vaccines and therapeutics manufacturing, plasma-derived blood products, and the growing market in cellular and gene therapy. Regulatory needs to perform multi-step viral clearance for all biological products ensure strong demand for filtration consumables in this segment. The fastest-growing part in the biologicals segment is in cellular and gene therapy due to scaling in viral vector production and commercialization in ex vivo cell therapy manufacturing platforms. Medical device applications are also growing in this market due to increasing stringency in validation needs for virus removal in device manufacturing. Water purification and air purification contribute incremental but stable growth in this market.
By Product, Consumables Lead the Market, While Services Register Fastest Growth
In 2025, the revenue share of consumables suffixed to the highest level of around 57.42 %, for the frequent demand of virus retentive membrane filters, kits & reagents and single use filtration assemblies as part of biologics production processes. Since batch process cycles often transcend capital equipment cycles in monoclonal antibody and plasma-derived product manufacturing processes, this high number of batch process cycles ensures constant consumable demand regardless of capital equipment cycles. However, the services segment is expected to show highest CAGR of 11.23% from 2026-2035, owing to the growing trend of outsourcing of viral clearance validation studies, filter performance qualification, and regulatory support services to contract testing organization (CTOs). Complex regulatory requirements as well as requirements for biosafety level-2 containment are exacerbating this trend toward third-party service models.
By Technology, Filtration Dominates, While Chromatography Shows Rapid Growth
The filtration technology segment had the highest share in terms of revenue, 61.85%, in 2025, due to the wide range of applicability in the manufacturing of biologics, blood fractionation, and plasma processing, with virus retentive nanofiltration membranes having high LRVs against enveloped and non-enveloped viruses. In addition, improvements in membrane chemistry, uniformity in pore size, and pre-validation in filter formats have helped this segment hold the highest place. The chromatography segment is expected to register the highest CAGR, approximately 11.47%, in the range from 2026 to 2035, due to the incorporation of this technology in biologic purification platforms as an orthogonal viral clearance step. Anion exchange and mixed-mode chromatography resin with flow-through viral clearance properties is seeing a surge in adoption in next-generation mAb and gene therapy purification platforms.
By End-Use, Biopharmaceutical and Biotechnology Companies Lead and Drive Sustained Demand
In 2025, the biopharm industry and biotechnology firms accounted for the biggest market share of about 54.71%. This has been driven by large-scale in-house production of biologics (and such a requirement to have validated viral clearance in GMP environments). These are also the largest purchasers of high dollar filter gear including assemblies and validation tools. Within this segment, the contract research organizations are projected to have the highest CAGR of about 11.84% during the forecast period (2023-2035). This is due to the increasing outsourcing of viral safety testing, clearance study development, and regulatory support to specialized CRO firms, as more pharmaceutical companies choose to conduct their research in this manner.
Virus Filtration Market Regional Highlights:
North America Virus Filtration Market Insights:
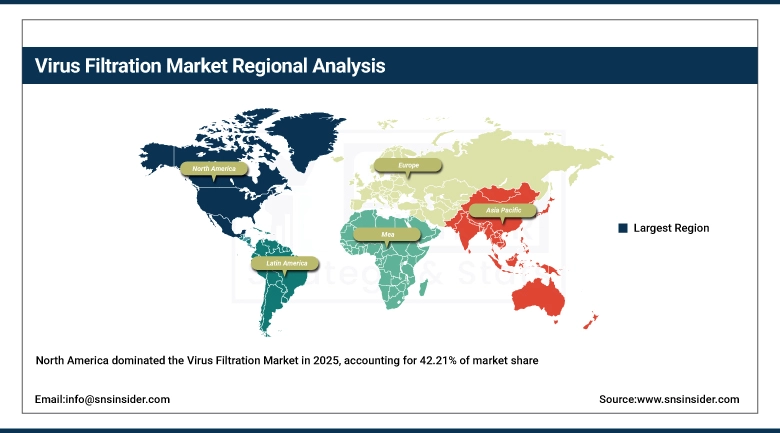
In 2025, the virus filtration market was dominated by North America, which held more than 42.21% of the total share. The reason for this however is its strong biopharma industry base; also due to the strong FDA CCV vaping safety testing, and high number of biologic (biologic) drug developers (including in the USA) and CDMOs. The mRNA vaccine development capacity, and the expansion of plasma fractionation capacity, combined with modernized Californian ATMP manufacturing facilities are projected to drive demand for virus-retentive filtration systems in the US, which represents a major commercial hub in North America. On top of that, Canada has seen a growth in biotech, significant global pharmaceutical headquarters reside in the US Northeast and West Coast corridors.
Get Customized Report as per Your Business Requirement - Enquiry Now
Asia Pacific Virus Filtration Market Insights:
The Asia Pacific region holds the highest growth potential for the virus filtration market, growing at a CAGR of 12.35%, owing to the increasing biopharmaceutical manufacturing capacity within the countries of China, India, South Korea, and Japan. Government initiatives such as the "14th Five-Year Plan" for biopharmaceutical manufacturing in China, the "National Biopharma Mission" initiative in India, and the "K-Bio strategy" initiative in South Korea are resulting in significant investment within the biopharmaceutical manufacturing industry. The increasing number of biosimilar companies within the region that are seeking regulatory approval within their respective countries as well as globally, the increasing vaccine production mandates within the region due to the COVID-19 pandemic, and the increasing number of CRO/CDMO companies within the region are all resulting in significant growth within the regional virus filtration market.
Europe Virus Filtration Market Insights:
The second-largest region for the virus filtration market is Europe. This region enjoys the advantages of EMA’s viral safety guidelines, existing plasma fractionation industries in Germany, France, and the UK, and an increasing advanced therapy pipeline. The guideline of the European Medicines Agency on virus safety evaluation, "CPMP/BWP/268/95," and the need to remove adventitious agents from biologics have fueled the need for high-performance virus filtration and chromatography systems within biopharma companies in Europe. Cross-border manufacturing harmonization under EU Pharma Legislation and biopharma clusters in Nordic and Eastern Europe are future growth opportunities.
Latin America (LATAM) and Middle East & Africa (MEA) Virus Filtration Market Insights:
Latin American adoption markets for virus filtration through national programs of biological manufacturing and the integration of increasing CDMO demand targeting regulated export markets. Key demand drivers include investments in infrastructure for plasma fractionation and vaccine manufacturing self-sufficiency. The growing demand for GMP-grade virus filtration consumables for their regional biologics production facilities positions the UAE and Saudi Arabia as up-and-coming regional biopharma manufacturing hubs fueled by burgeoning healthcare investment programs in the two nations aligned to Vision 2030 and the Middle East/Africa region. Another emerging trend in relation to basic plasma safety filtration investments is the increasing internet infrastructure in Sub-Saharan Africa.
Virus Filtration Market Competitive Landscape:
Merck KGaA (MilliporeSigma) (est. With an extensive portfolio covering Viresolve™ nanofiltration membranes, Millistak™ depth filters from Merck Group (Merck KGaA, Darmstadt, Germany), and single-use assemblies combined for world-class virus filtration solutions (1688), our trusted reputation as a leader in life science tools and bioprocess solutions takes us further ahead. Its extensive regulatory expertise and global application support network ensure the firm is the supplier of choice for the biologics manufacturers looking for scalable validated viral clearance solutions.
-
In February 2025, Merck KGaA launched an enhanced Viresolve Pro filtration platform with improved flux performance and a pre-validated, audit-ready viral clearance data package, designed to reduce regulatory submission timelines for biopharmaceutical manufacturers.
Sartorius AG, an international partner of the biopharmaceutical and laboratory industries, provides a full range of virus filtration products, from Virosart and Sartopore gamma-assisted components to filter capsules and single-use bioprocessing assemblies. Integrated Filtration Platform Development Sartorius applies hardware, software and consumables solution integration approach to provide platform solutions that cover the complete range of viral safety needs in biologics manufacture.
-
In September 2024, Sartorius AG expanded its BioStat STR bioreactor line with an integrated downstream viral filtration module, enabling seamless end-to-end bioprocessing from cell culture to viral clearance in a single automated platform for monoclonal antibody manufacturers.
Cytiva (est. as GE Healthcare Life Sciences, 2020 rebranded) is a prominent global provider of technologies and services that advance the discovery, manufacture, and delivery of therapeutics. Its Vivaflow and Ultipor VF viral removal filter product lines are widely used in plasma fractionation, vaccine manufacturing, and recombinant protein purification, and the company maintains deep integration with its AKTA chromatography and bioprocess automation platforms.
-
In April 2024, Cytiva introduced a next-generation Ultipor VF Grade DV20 filter with enhanced non-enveloped virus retention performance and a 30% improvement in filtrate flux, targeting high-throughput plasma and biologic purification applications.
Virus Filtration Market Key Players:
-
Merck KGaA
-
Sartorius AG
-
Cytiva
-
Pall Corporation
-
Thermo Fisher Scientific
-
Asahi Kasei Medical Co., Ltd.
-
Charles River Laboratories International
-
Lonza Group AG
-
Repligen Corporation
-
3M Purification Inc.
-
Parker Hannifin Corporation
-
Bio-Rad Laboratories, Inc.
-
Meissner Filtration Products, Inc.
-
WuXi Biologics
-
Entegris, Inc.
-
Cobetter Filtration Equipment Co., Ltd.
-
Novasep Holding SAS
-
Agilent Technologies, Inc.
-
Eurofins Scientific SE
-
Clean Harbors, Inc.
| Report Attributes | Details |
|---|---|
| Market Size in 2025 | USD 5.71 Billion |
| Market Size by 2035 | USD 15.00 Billion |
| CAGR | CAGR of 10.15% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Report Scope & Coverage | Market Size, Segments Analysis, Competitive Landscape, Regional Analysis, DROC & SWOT Analysis, Forecast Outlook |
| Key Segments | • By Product [Consumables (Kits and reagents, Others), Instruments (Filtration systems, Chromatography systems), Services] • By Technology [Filtration (Consumables, Instruments, Services), Chromatography (Consumables, Instruments, Services)] • By Application [Biologicals (Vaccines and therapeutics, Blood and blood products, Cellular and gene therapy products, Tissue and tissue products, Stem cell products), Medical devices, Water purification, Air purification] • By End-use [Biopharmaceutical & biotechnology companies, Contract research organizations, medical device companies, Academic institutes & research laboratories] |
| Regional Analysis/Coverage | North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, Poland, Turkey, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Singapore, Australia, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Rest of Latin America) |
| Company Profiles | Merck KGaA, Sartorius AG, Cytiva, Pall Corporation, Thermo Fisher Scientific, Asahi Kasei Medical Co., Ltd., Charles River Laboratories International, Lonza Group AG, Repligen Corporation, 3M Purification Inc., Parker Hannifin Corporation, Bio-Rad Laboratories, Inc., Meissner Filtration Products, Inc., WuXi Biologics, Entegris, Inc., Cobetter Filtration Equipment Co., Ltd., Novasep Holding SAS, Agilent Technologies, Inc., Eurofins Scientific SE, Clean Harbors, Inc. |
Frequently Asked Questions
North America leads the Virus Filtration Market with over 42.21% share in 2025, driven by advanced biopharma infrastructure and strong regulatory frameworks.
The Virus Filtration Market was valued at USD 5.71 billion in 2025, driven by rising biologics production and strict viral safety regulations.
The Virus Filtration Market is expected to reach USD 15.00 billion by 2035, reflecting strong long-term demand in biopharmaceutical manufacturing.
The Virus Filtration Market is projected to grow at a CAGR of 10.15% over the forecast period, supported by expanding cell and gene therapy pipelines.
Consumables dominate the Virus Filtration Market with a 57.42% share in 2025, due to recurring demand for filters, kits, and single-use systems.

